 PDF(1268 KB)
PDF(1268 KB)


Exercise Selections for People with Dementia for Prevention and Interventions
LiHao, LiuHao
Chinese Journal of Alzheimer's Disease and Related Disorders ›› 2021, Vol. 4 ›› Issue (2) : 156-162.
 PDF(1268 KB)
PDF(1268 KB)
Abbreviation (ISO4): Chinese Journal of Alzheimer's Disease and Related Disorders
Editor in chief: Jun WANG
 PDF(1268 KB)
PDF(1268 KB)
Exercise Selections for People with Dementia for Prevention and Interventions
With the increasing elderly population, the number of patients with Alzheimer's disease is also escalating which has caught attention worldwide. In addition to physical and psychological changes due to memory loss, the clinical symptoms of Alzheimer's disease are more important than the continuous decline in physical function and daily life ability, which affects the quality of life. Up to date there are effective no drugs for dementia, and exercise interventions is currently the most promising intervention to improve such patients' bone, muscle, heart, lung, and neural functions. However, how to choose and use different exercise therapy correctly needs to be further clarified in practice. In this prospective article, we demonstrate the effects of exercise therapy on physical function and quality of life in different stages of Alzheimer's disease from the perspective of prevention and intervention. It may provide valuable guidelines for rehabilitation professionals and caregivers to take good care of patients with dementia.
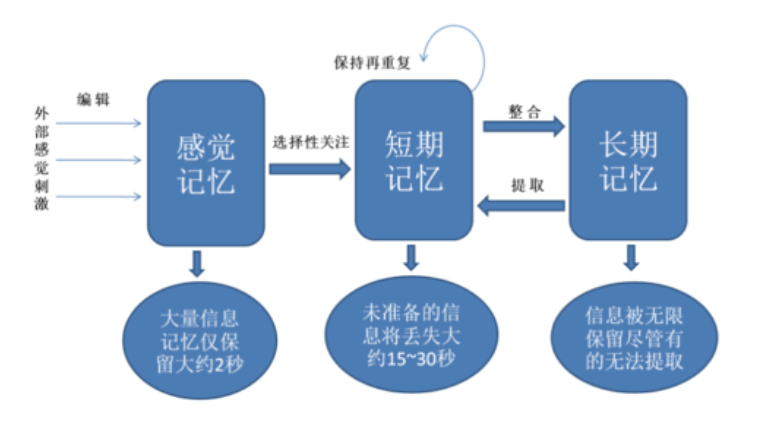
Dementia / Alzheimer disease / Physical exercises / function / Activities of Daily Living (ADL)
| [1] |
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
Exercise programs for people with dementia[J]. Cochrane Database Syst Rev, 2013,(12): CD006489.
|
| [12] |
The hippocampus shrinks in late adulthood, leading to impaired memory and increased risk for dementia. Hippocampal and medial temporal lobe volumes are larger in higher-fit adults, and physical activity training increases hippocampal perfusion, but the extent to which aerobic exercise training can modify hippocampal volume in late adulthood remains unknown. Here we show, in a randomized controlled trial with 120 older adults, that aerobic exercise training increases the size of the anterior hippocampus, leading to improvements in spatial memory. Exercise training increased hippocampal volume by 2%, effectively reversing age-related loss in volume by 1 to 2 y. We also demonstrate that increased hippocampal volume is associated with greater serum levels of BDNF, a mediator of neurogenesis in the dentate gyrus. Hippocampal volume declined in the control group, but higher preintervention fitness partially attenuated the decline, suggesting that fitness protects against volume loss. Caudate nucleus and thalamus volumes were unaffected by the intervention. These theoretically important findings indicate that aerobic exercise training is effective at reversing hippocampal volume loss in late adulthood, which is accompanied by improved memory function.
|
| [13] |
|
| [14] |
|
| [15] |
Background: Dementia affects cognitive functioning, physical functioning, activities of daily living (ADLs), and quality of life (QOL). Pharmacological treatments to manage, cure or prevent dementia remain controversial. Therefore development of non-pharmacological approaches to prevent, or at least delay the onset and progression of dementia is urgently needed. Passive exercise is proposed to be such a non-pharmacological alternative. This study primarily aims to investigate the effects of three different forms of passive exercise on QOL and ADLs of institutionalized patients with dementia. The secondary aims are to assess the effects of three different forms of passive exercise on cognitive functioning and physical functioning of institutionalized patients with dementia as well as on care burden of both the primary formal and primary informal caregivers of these patients.Methods: This is a multicenter randomized controlled trial. Three forms of passive exercise are distinguished; motion simulation (MSim), whole body vibration (WBV), and a combination of both MSim + WBV. Intervention effects are compared to a control group receiving regular care. Institutionalized patients with dementia follow a six-week intervention program consisting of four 4-12 min sessions a week.The primary outcome measures QOL and ADLs and secondary outcome measure care burden are assessed with questionnaires filled in by the primary formal and informal caregivers of the patient. The other secondary outcome measures cognitive and physical functioning are assessed by individual testing. The four groups are compared at baseline, after 6 weeks of intervention, and 2 weeks after the intervention has ended.Discussion: This study will provide insight in the effects of different forms of passive exercise on QOL, ADLs, cognitive and physical functioning and care burden of institutionalized patients with dementia and their primary formal and informal caregivers. The results of this study might support the idea that passive exercise can be an efficient alternative for physical activity for patients not able to be or stay involved in active physical exercise.
|
| [16] |
The differences in gait abnormalities from the earliest to the later stages of dementia and in the different subtypes of dementia have not been fully examined. This study aims to compare spatiotemporal gait parameters in cognitively healthy individuals, patients with amnestic mild cognitive impairment (MCI) and non-amnestic MCI, and patients with mild and moderate stages of Alzheimer's disease (AD) and non-Alzheimer's disease (non-AD).Based on a cross-sectional design, 1719 participants (77.4 ± 7.3 years, 53.9% female) were recruited from cohorts from seven countries participating in the Gait, Cognition and Decline (GOOD) initiative. Mean values and coefficients of variation of spatiotemporal gait parameters were measured during normal pace walking with the GAITRite system at all sites.Performance of spatiotemporal gait parameters declined in parallel with the stage of cognitive decline from MCI status to moderate dementia. Gait parameters of patients with non-amnestic MCI were more disturbed compared to patients with amnestic MCI, and MCI subgroups performed better than demented patients. Patients with non-AD dementia had worse gait performance than those with AD dementia. This degradation of gait parameters was similar between mean values and coefficients of variation of spatiotemporal gait parameters in the earliest stages of cognitive decline, but different in the most advanced stages, especially in the non-AD subtypes.Spatiotemporal gait parameters were more disturbed in the advanced stages of dementia, and more affected in the non-AD dementias than in AD. These findings suggest that quantitative gait parameters could be used as a surrogate marker for improving the diagnosis of dementia.© 2015 EAN.
|
| [17] |
|
| [18] |
|
| [19] |
The relationship between depression and dementia is complex and still not well understood. A number of different views exist regarding how the two conditions are linked as well as the underlying neurobiological mechanisms at work. This narrative review examined longitudinal and cross sectional studies in the existing literature and determined the evidence supporting depression being a risk factor, a prodrome, a consequence, or an independent comorbidity in dementia. Overall there is convincing evidence to support both the notion that early life depression can act as a risk factor for later life dementia, and that later life depression can be seen as a prodrome to dementia. There is also evidence to support both conditions showing similar neurobiological changes, particularly white matter disease, either indicating shared risk factors or a shared pattern of neuronal damage. These findings highlight the need to examine if effective treatment of depressive episodes has any effect in reducing the prevalence of dementia, as well as clinicians being vigilant for late life depression indicating the incipient development of dementia, and therefore carefully following up these individuals for future cognitive impairment. Copyright © 2014 Elsevier Ireland Ltd. All rights reserved.
|
| [20] |
|
| [21] |
The National Institute on Aging and the Office of Medical Applications of Research of the National Institutes of Health convened a State-of-the-Science Conference on 26-28 April 2010 to assess the available scientific evidence on prevention of cognitive decline and Alzheimer disease. This article provides the panel's assessment of the available evidence.
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
Background: With increasing age neuromuscular deficits (e.g., sarcopenia) may result in impaired physical performance and an increased risk for falls. Prominent intrinsic fall-risk factors are age-related decreases in balance and strength / power performance as well as cognitive decline. Additional studies are needed to develop specifically tailored exercise programs for older adults that can easily be implemented into clinical practice. Thus, the objective of the present trial is to assess the effects of a fall prevention program that was developed by an interdisciplinary expert panel on measures of balance, strength / power, body composition, cognition, psychosocial well-being, and falls self-efficacy in healthy older adults. Additionally, the time-related effects of detraining are tested.;Methods/Design: Healthy old people (n = 54) between the age of 65 to 80 years will participate in this trial. The testing protocol comprises tests for the assessment of static / dynamic steady-state balance (i.e., Sharpened Romberg Test, instrumented gait analysis), proactive balance (i.e., Functional Reach Test; Timed Up and Go Test), reactive balance (i.e., perturbation test during bipedal stance; Push and Release Test), strength (i.e., hand grip strength test; Chair Stand Test), and power (i.e., Stair Climb Power Test; countermovement jump). Further, body composition will be analysed using a bioelectrical impedance analysis system. In addition, questionnaires for the assessment of psychosocial (i.e., World Health Organisation Quality of Life Assessment-Bref), cognitive (i.e., Mini Mental State Examination), and fall risk determinants (i.e., Fall Efficacy Scale -International) will be included in the study protocol. Participants will be randomized into two intervention groups or the control / waiting group. After baseline measures, participants in the intervention groups will conduct a 12-week balance and strength / power exercise intervention 3 times per week, with each training session lasting 30 min. (actual training time). One intervention group will complete an extensive supervised training program, while the other intervention group will complete a short version (` 3 times 3') that is home-based and controlled by weekly phone calls. Post-tests will be conducted right after the intervention period. Additionally, detraining effects will be measured 12 weeks after program cessation. The control group / waiting group will not participate in any specific intervention during the experimental period, but will receive the extensive supervised program after the experimental period.;Discussion: It is expected that particularly the supervised combination of balance and strength / power training will improve performance in variables of balance, strength / power, body composition, cognitive function, psychosocial well-being, and falls self-efficacy of older adults. In addition, information regarding fall risk assessment, dose-response-relations, detraining effects, and supervision of training will be provided. Further, training-induced health-relevant changes, such as improved performance in activities of daily living, cognitive function, and quality of life, as well as a reduced risk for falls may help to lower costs in the health care system. Finally, practitioners, therapists, and instructors will be provided with a scientifically evaluated feasible, safe, and easy-to-administer exercise program for fall prevention.
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
Sampaio
|
| [35] |
|
/
| 〈 |
|
〉 |