 PDF(2641 KB)
PDF(2641 KB)


Analysis of changes in gut microbiota of 5XFAD transgenic mice
LUOZhi-yu, CHUXing-kun, CHANGShuai-shuai, FENGYuan, YANGDa-bing, WANGXin-yi, DUXiao-guang, FENGTeng, ZHANGJing
Chinese Journal of Alzheimer's Disease and Related Disorders ›› 2021, Vol. 4 ›› Issue (3) : 179-184.
 PDF(2641 KB)
PDF(2641 KB)
Abbreviation (ISO4): Chinese Journal of Alzheimer's Disease and Related Disorders
Editor in chief: Jun WANG
 PDF(2641 KB)
PDF(2641 KB)
Analysis of changes in gut microbiota of 5XFAD transgenic mice
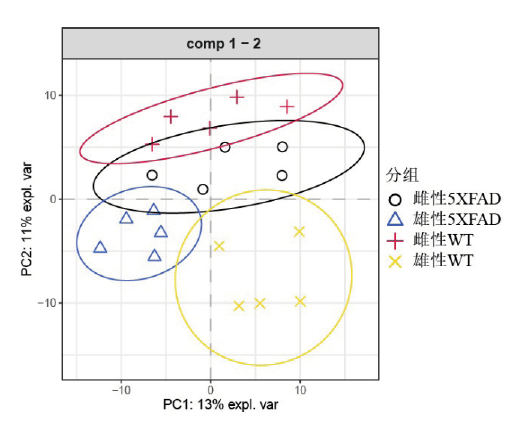
Objective: To explore the gut microbiota changes of 5XFAD transgenic mice of Alzheimer's disease (AD) model compared with wild-type (WT) mice and the impacts of gender difference. Methods: The feces of different gender 5XFAD and WT mice were collected, and the bacterial 16S rRNA of fecal genomic DNA was amplified by PCR and sequenced by using high-throughput sequencing methods. Then, the differential and functional analysis of the sequencing data are carried out through nf-core and the bioinformation analysis method based on the R language platform. Results: In the composition analysis, compared to WT mice, 5XFAD transgenic mice had lower abundance of Epsilonbacteraeota and Proteobacteria at the phylum level. At genus level, 5XFAD transgenic mice had higher abundance of Alistipes and Lactobacillus and lower abundance of Helicobacter. In the functional analysis, compared to WT mice, 5XFAD transgenic mice had influenced different metabolism pathways. Besides, the composition and function of microbiota was different in a gender specific manner. Conclusion: The gut microbiota of 5XFAD transgenic mice is different from that of WT mice, and the difference is affected by gender.
gut microbiota / 16S rRNA high-throughput sequencing / 5XFAD
| [1] |
Alzheimer's disease is a chronic illness with long preclinical and prodromal phases (20 years) and an average clinical duration of 8-10 years. The disease has an estimated prevalence of 10-30% in the population >65 years of age with an incidence of 1-3%. Most patients with Alzheimer's disease (>95%) have the sporadic form, which is characterized by a late onset (80-90 years of age), and is the consequence of the failure to clear the amyloid-β (Aβ) peptide from the interstices of the brain. A large number of genetic risk factors for sporadic disease have been identified. A small proportion of patients (<1%) have inherited mutations in genes that affect processing of Aβ and develop the disease at a much younger age (mean age of ∼45 years). Detection of the accumulation of Aβ is now possible in preclinical and prodromal phases using cerebrospinal fluid biomarkers and PET. Several approved drugs ameliorate some of the symptoms of Alzheimer's disease, but no current interventions can modify the underlying disease mechanisms. Management is focused on the support of the social networks surrounding the patient and the treatment of any co-morbid illnesses, such as cerebrovascular disease.
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
|
| [6] |
|
| [7] |
The human gut harbors diverse microbes that play a fundamental role in the well-being of their host. The constituents of the microbiota--bacteria, viruses, and eukaryotes--have been shown to interact with one another and with the host immune system in ways that influence the development of disease. We review these interactions and suggest that a holistic approach to studying the microbiota that goes beyond characterization of community composition and encompasses dynamic interactions between all components of the microbiota and host tissue over time will be crucial for building predictive models for diagnosis and treatment of diseases linked to imbalances in our microbiota.Copyright © 2012 Elsevier Inc. All rights reserved.
|
| [8] |
|
| [9] |
There is a growing emphasis on the relationship between the microorganisms inhabiting the gut (gastrointestinal microbiota) and human health. The emergence of a microbiota-gut-brain axis to describe the complex networks and relationship between the gastrointestinal microbiota and host reflects the major influence this environment may have in brain health and disorders of the central nervous system (CNS). Bidirectional communication between the microbiota and the CNS occurs through autonomic, neuroendocrine, enteric, and immune system pathways. Potential neurobiological mechanisms through which disruptions in this network may impact health and disease include hypothalamic-pituitary-adrenal (HPA)-axis activation, and altered activity of neurotransmitter and immune systems. Perturbations of the gut microbial community have already been implicated in multiple host diseases such as obesity, diabetes, and inflammation, while recent evidence suggests a potential role of the microbiota-gut-brain axis in neuropsychiatric disorders, such as depression and anxiety. Here, we review the current literature related to the influence of the gut microbial community on central nervous system function, with a specific focus on anxiety and depressive symptoms. The role of stress and stress-mediated changes in autonomic, neuroendocrine, immune, and neurotransmitter systems are examined, followed by a discussion of the role of the microbiota in novel gastrointestinal-based treatment options for the prevention and treatment of brain-based disorders such as anxiety and depression.Copyright © 2017 Elsevier Inc. All rights reserved.
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
Recent evidence suggests the commensal microbiome regulates host immunity and influences brain function; findings that have ramifications for neurodegenerative diseases. In the context of Alzheimer's disease (AD), we previously reported that perturbations in microbial diversity induced by life-long combinatorial antibiotic (ABX) selection pressure in the APP/PS1 mouse model of amyloidosis is commensurate with reductions in amyloid-β (Aβ) plaque pathology and plaque-localised gliosis. Considering microbiota-host interactions, specifically during early post-natal development, are critical for immune- and neuro-development we now examine the impact of microbial community perturbations induced by acute ABX exposure exclusively during this period in APP/PS1 mice. We show that early post-natal (P) ABX treatment (P14-P21) results in long-term alterations of gut microbial genera (predominantly Lachnospiraceae and S24-7) and reduction in brain Aβ deposition in aged APP/PS1 mice. These mice exhibit elevated levels of blood- and brain-resident Foxp3 T-regulatory cells and display an alteration in the inflammatory milieu of the serum and cerebrospinal fluid. Finally, we confirm that plaque-localised microglia and astrocytes are reduced in ABX-exposed mice. These findings suggest that ABX-induced microbial diversity perturbations during post-natal stages of development coincide with altered host immunity mechanisms and amyloidosis in a murine model of AD.
|
| [18] |
Alzheimer's disease is the most common form of dementia in the western world, however there is no cure available for this devastating neurodegenerative disorder. Despite clinical and experimental evidence implicating the intestinal microbiota in a number of brain disorders, its impact on Alzheimer's disease is not known. To this end we sequenced bacterial 16S rRNA from fecal samples of Aβ precursor protein (APP) transgenic mouse model and found a remarkable shift in the gut microbiota as compared to non-transgenic wild-type mice. Subsequently we generated germ-free APP transgenic mice and found a drastic reduction of cerebral Aβ amyloid pathology when compared to control mice with intestinal microbiota. Importantly, colonization of germ-free APP transgenic mice with microbiota from conventionally-raised APP transgenic mice increased cerebral Aβ pathology, while colonization with microbiota from wild-type mice was less effective in increasing cerebral Aβ levels. Our results indicate a microbial involvement in the development of Abeta amyloid pathology, and suggest that microbiota may contribute to the development of neurodegenerative diseases.
|
| [19] |
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
|
/
| 〈 |
|
〉 |