 PDF(1502 KB)
PDF(1502 KB)


Quantifying Extrapyramidal Iron Deposition in Patients with Alzheimer's disease using Quantitative Susceptibility Mapping
DONGJun-yi, LIUYang-ying-qiu, GAOBing-bing, TIANShi-yun, SONGQing-wei, MIAOYan-wei
Chinese Journal of Alzheimer's Disease and Related Disorders ›› 2021, Vol. 4 ›› Issue (3) : 231-236.
 PDF(1502 KB)
PDF(1502 KB)
Abbreviation (ISO4): Chinese Journal of Alzheimer's Disease and Related Disorders
Editor in chief: Jun WANG
 PDF(1502 KB)
PDF(1502 KB)
Quantifying Extrapyramidal Iron Deposition in Patients with Alzheimer's disease using Quantitative Susceptibility Mapping
Objective: To quantitative the iron deposition of extrapyramidal nuclei in patients with Alzheimer's disease (AD) by quantitative susceptibility mapping(QSM) and to analyze the correlation between magnetic sensitivity values(MSV) and clinical laboratory indicators and cognitive scores. Methods: Fifty-nine patients with AD (AD group) and 22 aged volunteers without cognitive impairment (CON group) underwent routine MRI and ESWAN examination. SPIN software was used for image post-processing. MSV of bilateral head of caudate nucleus(HCN), globus pallidus(GP), putamen(PUT), thalamus(THA), red nucleus(RN), substantia nigra(SN) and dentate nucleus(DN) were measured respectively, and correlation analysis was conducted with cognitive scores and general data. The area under ROC curve was used to evaluate the effectiveness of MSV values in diagnosing AD. Results: The MSV of all nuclei in AD group were higher than CON group, and there were significant differences in bilateral HCN, bilateral GP, bilateral THA, right RN, right SNand left DN (P< 0.05). The MSV of bilateral HCN and bilateral PUT had a significant negative correlation with the MMSE score. There was a significant negative correlation between the MSV of bilateral HCN, bilateral PUT and MoCA scores. The MSV of bilateral HCN, bilateral PUT had a significant negative correlation with CDT scores (all P< 0.05). The MSV of right GP was positively correlated with the course of the disease; The MSV of bilateral THA were inversely related to triglyceride content(all P< 0.05). ROC analysis showed that MSV of the left GP had the largest AUC. Conclusion: Increasing iron deposition of extrapyramidal nuclei in AD patients may affect cognitive status.
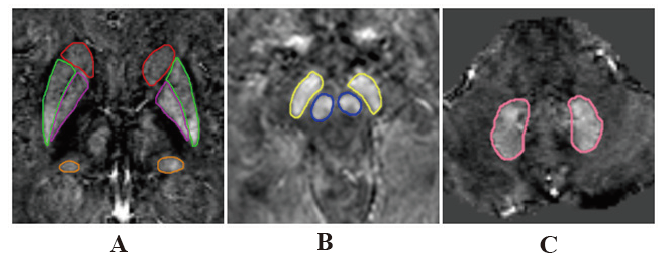
Alzheimer's disease / extrapyramidal / iron deposition / quantitative magnetic susceptibility / magnetic sensitivity values
| [1] |
|
| [2] |
Iron is the most abundant trace element in the human body. It is well known that iron is an important component of hemoglobin involved in the transport of oxygen. As a component of various enzymes, it participates in the tricarboxylic acid cycle and oxidative phosphorylation. Iron in the nervous system is also involved in the metabolism of catecholamine neurotransmitters and is involved in the formation of myelin. Therefore, iron metabolism needs to be strictly regulated. Previous studies have shown that iron deficiency in the brain during infants and young children causes mental retardation, such as delayed development of language and body balance, and psychomotor disorders. However, if the iron is excessively deposited in the aged brain, it is closely related to the occurrence of various neurodegenerative diseases, such as Alzheimer's disease, Parkinson's disease, and Friedreich's ataxia. Therefore, it is important to fully study and understand the mechanism of brain iron metabolism and its regulation. On this basis, exploring the relationship between brain iron regulation and the occurrence of nervous system diseases and discovering new therapeutic targets related to iron metabolism have important significance for breaking through the limitation of prevention and treatment of nervous system diseases. This review discusses the complete research history of iron and its significant role in the pathogenesis of the central nervous system (CNS) diseases.
|
| [3] |
|
| [4] |
In MRI, the main magnetic field polarizes the electron cloud of a molecule, generating a chemical shift for observer protons within the molecule and a magnetic susceptibility inhomogeneity field for observer protons outside the molecule. The number of water protons surrounding a molecule for detecting its magnetic susceptibility is vastly greater than the number of protons within the molecule for detecting its chemical shift. However, the study of tissue magnetic susceptibility has been hindered by poor molecular specificities of hitherto used methods based on MRI signal phase and T2* contrast, which depend convolutedly on surrounding susceptibility sources. Deconvolution of the MRI signal phase can determine tissue susceptibility but is challenged by the lack of MRI signal in the background and by the zeroes in the dipole kernel. Recently, physically meaningful regularizations, including the Bayesian approach, have been developed to enable accurate quantitative susceptibility mapping (QSM) for studying iron distribution, metabolic oxygen consumption, blood degradation, calcification, demyelination, and other pathophysiological susceptibility changes, as well as contrast agent biodistribution in MRI. This paper attempts to summarize the basic physical concepts and essential algorithmic steps in QSM, to describe clinical and technical issues under active development, and to provide references, codes, and testing data for readers interested in QSM.© 2014 The Authors. Magnetic Resonance in Medicine Published by Wiley Periodicals, Inc. on behalf of International Society of Medicine in Resonance.
|
| [5] |
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
To quantify iron deposition in Alzheimer's disease (AD), amnestic mild cognitive impairment (aMCI), and control individuals using susceptibility weighted imaging (SWI).Sixty participants (22 aMCI, 20 AD, 18 normal controls) underwent conventional magnetic resonance imaging (MRI) and SWI using axial/oblique coronal sequences. Phase images were used to calculate bilateral iron deposition in 18 regions of interest (ROI). The radian angle value was calculated and compared between the three participant groups.The difference in radian angle value was significant between the aMCI and control groups in the left (L)-hippocampus, L-head of the caudate nucleus, R-lenticular nucleus, L-lenticular nucleus (P =0.02239, <0. 001, 0.03571, 0.00943, respectively). The difference in radian angle value was significant between the AD and aMCI groups in the R-cerebellar hemisphere, L-cerebellar hemisphere, R-hippocampus, L-hippocampus, R-red nucleus, R-thalamus, L-thalamus, and splenium of corpus callosum (P =0.02754, 0.01839, 0.00934, 0.04316, 0.02472, 0.00152, <0.001, 0.01448, respectively). Pearson correlation coefficients of the Mini-Mental State Examination score were all significant for the bilateral cerebellar hemisphere, hippocampus, red nucleus, lenticular nucleus, thalamus, R-head of the caudate nucleus, and splenium of corpus callosum.Iron deposition in the hippocampus, head of the caudate nucleuslenticular nucleus, and thalamus are significantly different between individuals with aMCI, AD, and controls. The thalamus is a particularly sensitive area. Using SWI to quantify the iron deposition is a useful tool in detecting aMCI and AD.
|
| [18] |
|
| [19] |
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
Abnormal iron distribution in the isocortex is increasingly recognized as an in vivo marker for Alzheimer's disease (AD). However, the contribution of iron accumulation to the AD pathology is still poorly understood. In this study, we investigated: 1) frontal cortical iron distribution in AD and normal aging and 2) the relation between iron distribution and degree of AD pathology. We used formalin fixed paraffin embedded frontal cortex from 10 AD patients, 10 elder, 10 middle aged, and 10 young controls and visualized iron with a modified Perl's histochemical procedure. AD and elderly subjects were not different with respect to age and sex distribution. Iron distribution in the frontal cortex was not affected by normal aging but was clearly different between AD and controls. AD showed accumulation of iron in plaques, activated microglia, and, in the most severe cases, in the mid-cortical layers along myelinated fibers. The degree of altered iron accumulations was correlated to the amount of amyloid-β plaques and tau pathology in the same block, as well as to Braak stage (p < 0.001). AD and normal aging show different iron and myelin distribution in frontal cortex. These changes appear to occur after the development of the AD pathological hallmarks. These findings may help the interpretation of high resolution in vivo MRI and suggest the potential of using changes in iron-based MRI contrast to indirectly determine the degree of AD pathology in the frontal cortex.
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
This study aimed to investigate age-related iron deposition changes in healthy subjects and Alzheimer disease patients using susceptibility weighted imaging. The study recruited 182 people, including 143 healthy volunteers and 39 Alzheimer disease patients. All underwent conventional magnetic resonance imaging and susceptibility weighted imaging sequences. The groups were divided according to age. Phase images were used to investigate iron deposition in the bilateral head of the caudate nucleus, globus pallidus and putamen, and the angle radian value was calculated. We hypothesized that age-related iron deposition changes may be different between Alzheimer disease patients and controls of the same age, and that susceptibility weighted imaging would be a more sensitive method of iron deposition quantification. The results revealed that iron deposition in the globus pallidus increased with age, up to 40 years. In the head of the caudate nucleus, iron deposition peaked at 60 years. There was a general increasing trend with age in the putamen, up to 50-70 years old. There was significant difference between the control and Alzheimer disease groups in the bilateral globus pallidus in both the 60-70 and 70-80 year old group comparisons. In conclusion, iron deposition increased with age in the globus pallidus, the head of the caudate nucleus and putamen, reaching a plateau at different ages. Furthermore, comparisons between the control and Alzheimer disease group revealed that iron deposition changes were more easily detected in the globus pallidus. Crown Copyright © 2014. Published by Elsevier Ireland Ltd. All rights reserved.
|
/
| 〈 |
|
〉 |