 PDF(22847 KB)
PDF(22847 KB)


 PDF(22847 KB)
PDF(22847 KB)
 PDF(22847 KB)
PDF(22847 KB)
全固态钠离子电池及界面改性
All Solid-State Sodium Batteries and Its Interface Modification
全固态钠离子电池具有原料成本低、安全性高以及能量密度高等特点,在移动电源、电动汽车和大规模储能系统领域表现出巨大的应用潜力。然而全固态钠离子电池的发展和规模化应用亟需解决固体电解质室温离子电导率低、界面电荷转移阻抗大、固体电解质与电极界面兼容性和接触差等问题。本文结合近年来全固态钠离子电池相关报道和本课题组研究成果,概述了β-Al2O3型固体电解质、NASICON型固体电解质、硫化物固体电解质、聚合物固体电解质、复合固体电解质的研究进展及发展趋势;综述了全固态钠离子电池界面特性、固体电解质表面修饰、电极/固体电解质界面改性最新研究成果;最后对全固态钠离子电池界面改性策略发展方向进行了展望。本综述有助于加深对全固态钠离子电池界面科学问题的认识,并对固态钠离子电池的发展应用形成理论指导。
All solid-state sodium batteries have great potential for portable electronics, electric vehicles, and large-scale energy storage applications due to the low cost of sodium, high security, and high energy density. However, the development and large-scale application of all-solid-state sodium ion batteries urgently need to solve the problems such as low ion conductivity of solid electrolyte, high charge-transfer impedance on interface, insufficient interfacial contact, and compatibility issues between electrodes and electrolytes solid electrolyte. Herein, combining the latest reports with our research findings, the research progress and development trend of β-Al2O3 electrolytes, NASICON electrolytes, sulfide electrolytes, polymer electrolytes, and composite electrolytes were summarized. The latest achievements in interface characteristics, the modification strategies of the interface between the electrodes and solid electrolytes and modification methods for surfaces of solid electrolytes were reviewed. Finally, the development direction of interface modification strategy for solid-state sodium ion batteries was prospected. This review have contributed to understand the interface science issues of all solid-state sodium ion batteries and provides a theoretical guidance for the development and application of solid-state sodium ion batteries.
1 Introduction
2 Solid-state electrolytes
3 Challenges for all solid-state sodium batteries
4 Interfaces engineering
4.1 Cathode/electrolyte interfaces
4.2 Anode/electrolytes interfaces
4.3 Structure design for interfaces engineering
5 Conclusion and future perspectives
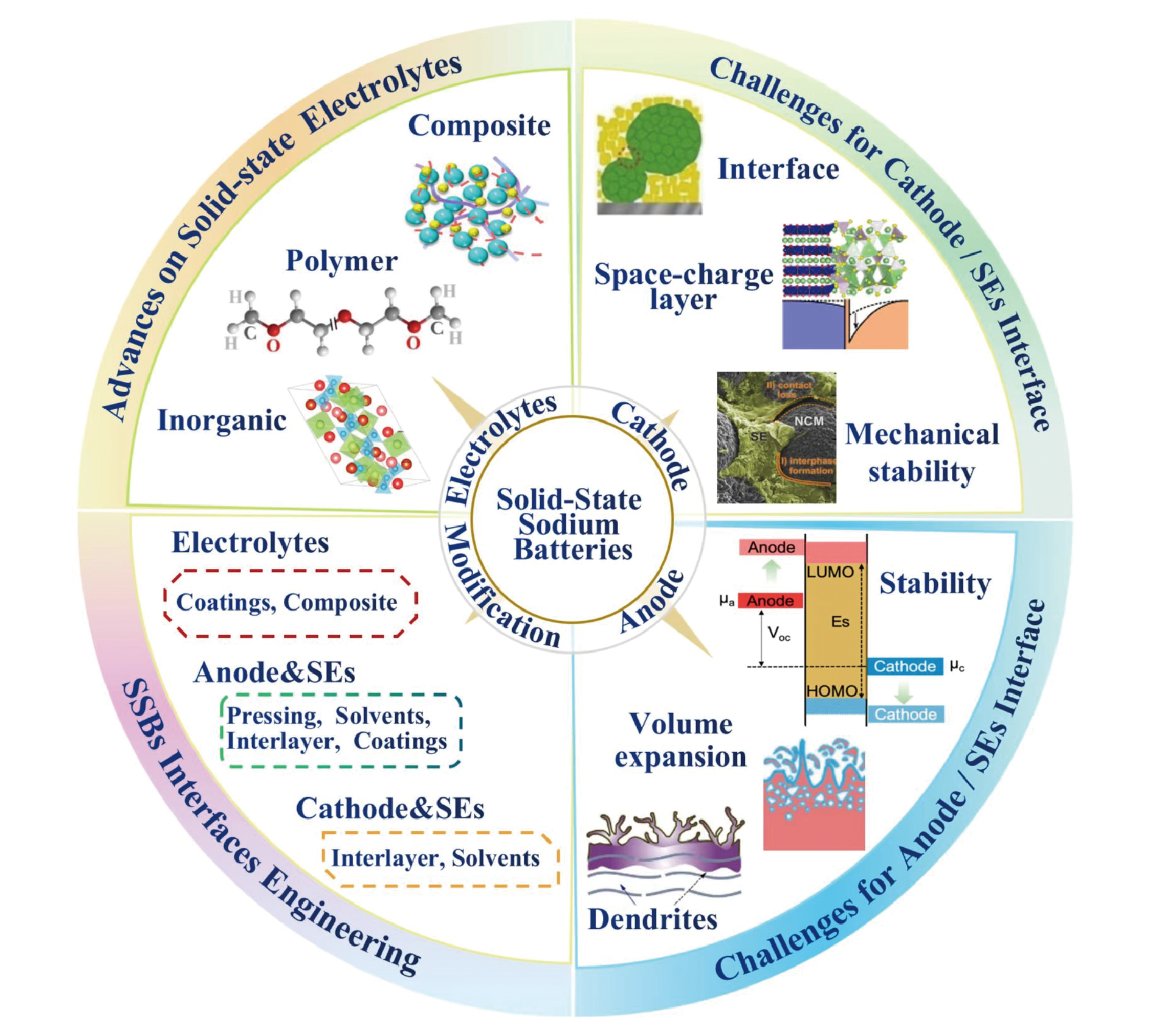
all solid-state sodium batteries / solid-state electrolytes / interface / modification
| [1] |
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
|
| [19] |
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
|
| [36] |
|
| [37] |
|
| [38] |
|
| [39] |
|
| [40] |
|
| [41] |
|
| [42] |
|
| [43] |
|
| [44] |
|
| [45] |
|
| [46] |
|
| [47] |
|
| [48] |
|
| [49] |
|
| [50] |
|
| [51] |
|
| [52] |
(张庆凯, 梁风, 姚耀春, 马文会, 杨斌, 戴永年. 化学进展, 2019, 31(1): 210.).
|
| [53] |
|
| [54] |
|
| [55] |
|
| [56] |
|
| [57] |
|
| [58] |
|
| [59] |
|
| [60] |
|
| [61] |
|
| [62] |
|
| [63] |
|
| [64] |
|
| [65] |
|
| [66] |
|
| [67] |
|
| [68] |
|
| [69] |
|
| [70] |
|
| [71] |
|
| [72] |
|
| [73] |
|
| [74] |
|
| [75] |
|
| [76] |
|
| [77] |
|
| [78] |
|
| [79] |
|
| [80] |
|
| [81] |
|
| [82] |
|
| [83] |
|
| [84] |
|
| [85] |
|
| [86] |
|
| [87] |
|
| [88] |
|
| [89] |
|
| [90] |
|
| [91] |
|
| [92] |
|
| [93] |
|
| [94] |
|
| [95] |
|
| [96] |
|
| [97] |
|
| [98] |
|
| [99] |
|
| [100] |
|
| [101] |
|
| [102] |
(陈龙, 黄少博, 邱景义, 张浩, 曹高萍. 化学进展, 2021, 33(8): 1378.).
|
| [103] |
|
| [104] |
|
| [105] |
|
| [106] |
|
| [107] |
|
| [108] |
|
| [109] |
|
| [110] |
|
| [111] |
|
| [112] |
|
| [113] |
|
| [114] |
|
| [115] |
|
/
| 〈 |
|
〉 |