 PDF(24225 KB)
PDF(24225 KB)


Metal-Support Interactions for Electrocatalytic Water Splitting
Aojie Yuan, Huan Liu, Danyang Hu, Lin Lan, Long Chen
Prog Chem ›› 2026, Vol. 38 ›› Issue (3) : 443-464.
 PDF(24225 KB)
PDF(24225 KB)
 PDF(24225 KB)
PDF(24225 KB)
Metal-Support Interactions for Electrocatalytic Water Splitting
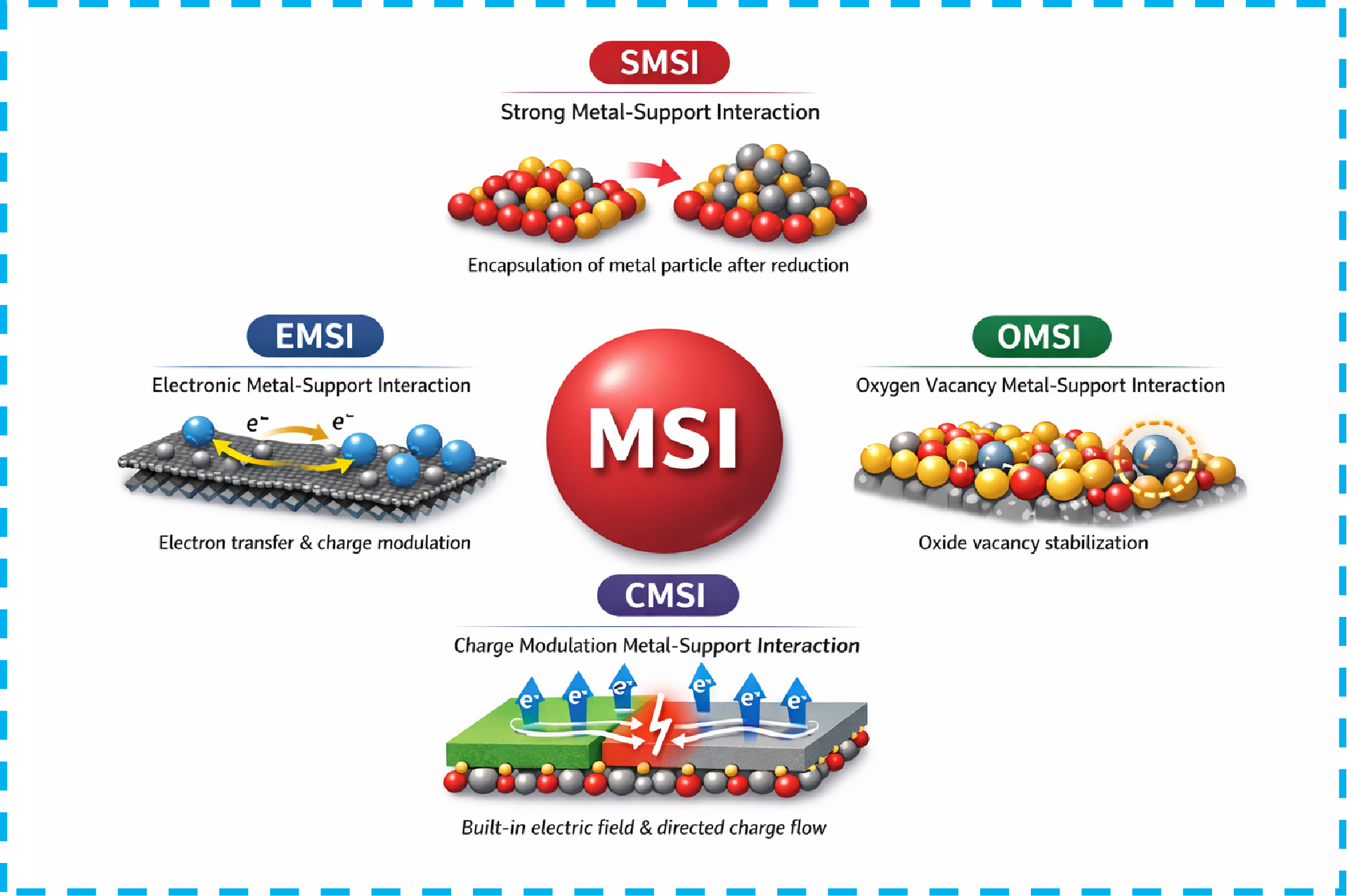
Metal-support interactions (MSIs) strategy play a critical role in designing and optimizing water-splitting catalysts. This review constructs a comprehensive framework for MSIs research, spanning from theoretical foundations to water-splitting applications. The fundamental concepts and historical evolution of MSIs are clarified, together with a taxonomic classification based on their physicochemical nature. On this basis, it delves into the formation mechanisms of various MSIs and systematically summarizes advanced characterization techniques used to analyze their electronic structures and interfacial properties. This review further explores how support properties, metal morphology, and preparation conditions collectively determine the strength and interaction mode of MSIs. A dedicated section introduces enhancement strategies, summarizing recent approaches for strengthening MSIs effects through defect engineering, interfacial design, and dynamic regulation. The applications of MSIs regulation in hydrogen evolution reaction (HER), oxygen evolution reaction (OER), and overall water splitting systems (OWS) are comprehensively discussed, along with the corresponding activity-enhancement mechanisms. It also outlines the challenges and future development directions in this field concerning atom-level precision control, operational condition characterization, and large-scale application.
1 Introduction
2 The formation and classification of MSIs
2.1 The formation of MSIs
2.2 The classification of MSIs
3 Characterization of MSIs
3.1 XANES
3.2 AC-TEM
3.3 Density functional theory of MSIs
3.4 Others
4 The formation and influencing factors of MSIs
4.1 Support
4.2 Metal
4.3 Interface temperature effect
5 Application of MSIs in HER and OER
5.1 HER
5.2 OER
6 Summary and outlook
6.1 Summary
6.2 Outlook
| [1] |
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
|
| [19] |
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
|
| [36] |
|
| [37] |
|
| [38] |
|
| [39] |
|
| [40] |
|
| [41] |
|
| [42] |
|
| [43] |
|
| [44] |
|
| [45] |
|
| [46] |
|
| [47] |
|
| [48] |
|
| [49] |
|
| [50] |
|
| [51] |
|
| [52] |
|
| [53] |
|
| [54] |
|
| [55] |
|
| [56] |
|
| [57] |
|
| [58] |
|
| [59] |
|
| [60] |
|
| [61] |
|
| [62] |
|
| [63] |
|
| [64] |
|
| [65] |
|
| [66] |
|
| [67] |
|
| [68] |
|
| [69] |
|
| [70] |
|
| [71] |
|
/
| 〈 |
|
〉 |