

Biomass to High Performance Adhesives: A New Strategy for Green Bonding
Received date: 2025-03-06
Revised date: 2025-04-10
Online published: 2025-09-01
Supported by
The National Natural Science Foundation of China(22278049)
The National Natural Science Foundation of China(U24A20559)
The Dalian High-Level Talent Innovation Program(2024RJ017)
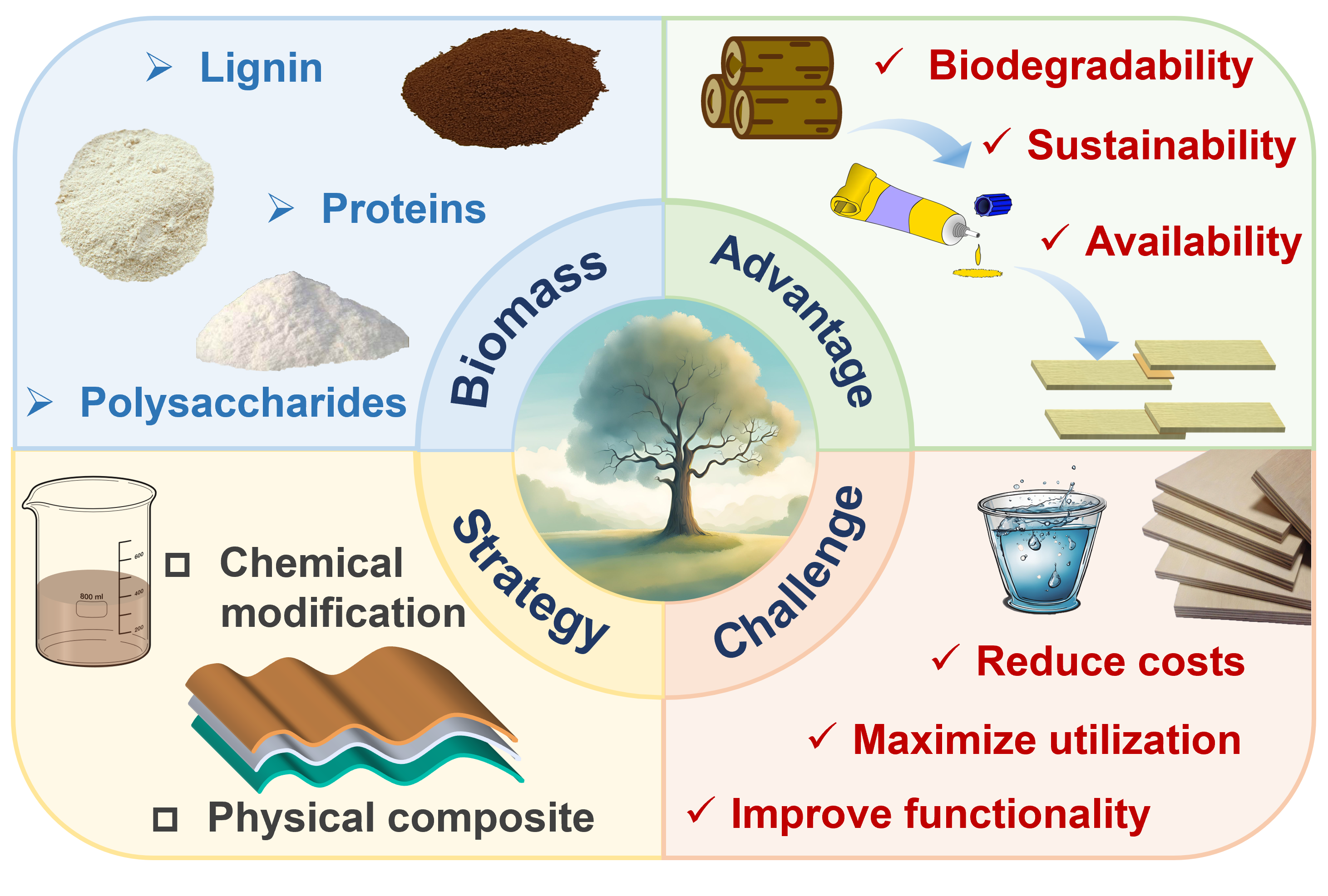
In the era of heightened global environmental consciousness, the principle of sustainable development has become deeply ingrained in public awareness. However, conventional petroleum-based adhesives are plagued by issues of unsustainability, high energy consumption, and significant environmental pollution during their production and application. Consequently, the development of green, sustainable, and high-performance biomass-based adhesives has emerged as a critical research focus. Biomass-based adhesives continue to encounter significant challenges, including suboptimal water resistance, elevated production costs, and the necessity for enhanced environmental performance. Future research should focus on optimizing the modification process of biomass raw materials, reducing production costs, improving the comprehensive properties of adhesives, and promoting their large-scale industrial application. In-depth investigation into the correlation between the structure and properties of biomass is crucial for the development of environmentally friendly and cost-effective adhesives. This paper summarizes the classification, modification methods, and properties of biomass-based raw materials and provides a detailed prospect for their future development.
1 Introduction
2 Modification strategies for the preparation of bio-based adhesives
2.1 Physical modification
2.2 Chemical modification
2.3 Composite modification
3 Adhesive production from biomass-based material
3.1 Lignin
3.2 Polysaccharides
3.3 Proteins
4 Conclusion and outlook
Key words: biomass; lignin; polysaccharides; proteins; adhesives; sustainable development
Zou Shuanglin , Xu Yingchun , Gui Tao , Tan Rong , Xiao Lingping , Sun Runcang . Biomass to High Performance Adhesives: A New Strategy for Green Bonding[J]. Progress in Chemistry, 2025 , 37(9) : 1352 -1360 . DOI: 10.7536/PC20250305
图1 氨基酸(AA)接枝结合脱甲基化修饰赋予碱木质素(AL)类贻贝黏附性,通过贻贝黏附蛋白中存在的离子-π/空间相关相互作用实现分子内协同作用[25]Fig.1 Amino acid (AA) grafting combining demethylating modification endows alkali lignin (AL) with mussel-like adhesion, realizing intramolecular synergy via ion-π/spatial correlation interactions existing in mussel adhesive proteins[25] |
图3 (a) 合成木质素-酚-甲醛(LPF)树脂胶黏剂(木质素、苯酚和甲醛的水溶液)的传统工艺; (b) 无酚木质素-甲醛(LF)胶黏剂(木质素的水悬浮液)的合成工艺[28]Fig.3 (a) Traditional process for synthesis of lignin-phenol-formaldehyde (LPF) resin adhesives (an aqueous solution of lignin, phenol, and formaldehyde). (b) The proposed process for synthesis of phenol-free lignin-formaldehyde (LF) adhesives (an aqueous suspension of lignin) in this study[28] |
图5 (a) 用稳定的酯键取代弱氢键; (b) 化学交联淀粉胶黏剂的黏合强度直方图; (c) 比较不同胶黏剂黏合强度直方图; (d) 本研究使用生态胶黏剂与先前确定的胶黏剂检查胶合板的黏合强度、低毒性和生物质含量[31]Fig.5 (a) Replacement of weak hydrogen bonds by stable ester bonds; (b) The bonding strength histogram of chemical cross-linking starch adhesives; (c) Histogram comparing the adhesive strength of different adhesives; (d) This study examines the bonding strength, hypo toxicity, and biomass contents of plywood using ecological adhesives to previously identified adhesives[31] |
图6 (a) 不同温度下处理的GTA-h凝胶和GTA-h70凝胶在不同底物上的黏附强度; (b) GTA凝胶在25 °C和60 ℃的冷热循环试验中G′和tanδ的变化以及在10个重复循环中对应的水下黏附强度; (c) GTA-h60水凝胶在水下黏附不同材料的照片[33]Fig.6 (a) Adhesion strength of the GTA-h gel treated at different temperatures and the GTA-h70 gel on different substrates. (b) The change of G′ and tanδ of the GTA gel during the heating-cooling cyclic tests (25 °C and 60 °C) and the corresponding underwater adhesion strength during ten repeated cycles. (c) Photos of the GTA-h60 hydrogel underwater adhered with different materials[33] |
图7 (a) CA/TA/FC多功能纳米球的制备流程图; (b) TA与FC之间相互作用; (c) HNTs@SH制备示意图; (d) SM-CA/TA/FC-HNTs@SH制备流程图[36]Fig.7 (a) Flow diagram of CA/TA/FC multifunctional nanospheres preparation. (b) The interactions between TA and FC. (c) Schematic diagram of HNTs@SH preparation. (d) Flow diagram of SM-CA/TA/FC-HNTs@SH preparation[36] |
表1 近年来报道的生物质基原料制备胶黏剂Table 1 Recently reported adhesives prepared from biomass-based raw materials |
| Material | Shear strength (MPa) | Ref |
|---|---|---|
| Alkali lignin | 17.00 | 11 |
| Lignin-glyoxal | 3.90 | 37 |
| Acetonitrile extraction of lignin | 1.70 | 10 |
| Lignosulfonate | 0.46 | 26 |
| Castor oil based polyurethane | 9.68 | 4 |
| Dialdehyde cellulose | 2.74 | 30 |
| Microcrystalline cellulose | 6.02 | 8 |
| Cellulose | 1.71 | 9 |
| Aminated cellulose | 2.56 | 38 |
| Cellulose nanocrystals | 29.00 | 39 |
| Starch | 1.87 | 7 |
| Starch/tannic acid | 1.73 | 40 |
| Starch/gelatin | 6.46 | 32 |
| Starch/Citric acid | 1.46 | 41 |
| Vegetable protein | 1.21 | 14 |
| Gelatin | 2.64 | 42 |
| Eugenol/epoxy resin | 19.80 | 43 |
| [1] |
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
|
| [19] |
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
|
| [36] |
|
| [37] |
|
| [38] |
|
| [39] |
|
| [40] |
|
| [41] |
|
| [42] |
|
| [43] |
|
/
| 〈 |
|
〉 |