

The Treatment and Disposal of Radioactive Liquid Waste and Solid Waste in Nuclear Power Plants
Received date: 2025-01-26
Revised date: 2025-03-31
Online published: 2025-09-01
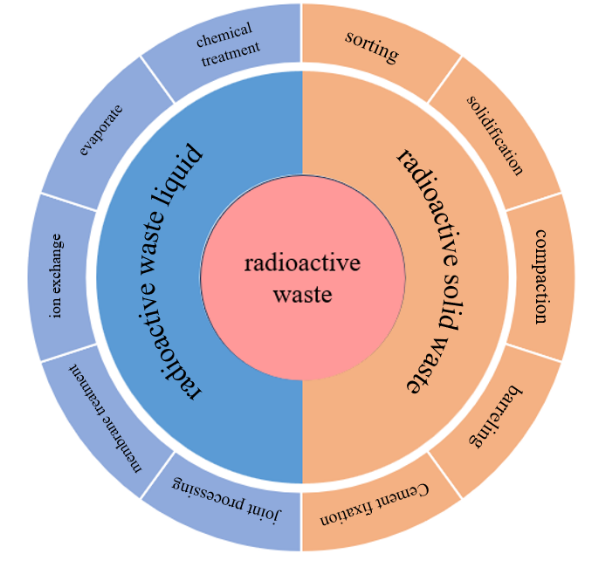
The increasing proportion of nuclear energy in China’s energy resources has brought about a series of difficulties and challenges. Nuclear power plants generate a large amount of radioactive liquid and solid waste during operation, and how to effectively treat and dispose of them has become a research focus. For radioactive liquid waste, the current main treatment processes in China are ion exchange and barrel evaporation drying. In addition, chemical precipitation, membrane technology, and other emerging technologies are also the current research directions for combined treatment. For solid waste, radioactive ions are tightly bound to solid materials, making it difficult for decontamination and regulatory release. Currently, solidification and compression are used for disposal in China, especially for mixed waste resins, which have large output and high radiation dose, as well as water absorption and elasticity, the main method in China is to use hot state overpressure technology to improve the volume reduction ratio, and then package and dispose of it geologically.
1 Background
2 Treatment of radioactive liquid waste
2.1 Radioactive wastewater
2.2 Treatment methods of radioactive liquid waste
3 Disposal of radioactive solid waste
4 Summary
4.1 Treatment technologies for liquid waste
4.2 Disposal of solid waste
5 Prospect
5.1 Research directions for the treatment of radioactive liquid waste
5.2 Research directions for radioactive solid waste
Yang Jingyuan , Yao Xiaoqi , Ye Li , Jin Hairui , Wang Yi . The Treatment and Disposal of Radioactive Liquid Waste and Solid Waste in Nuclear Power Plants[J]. Progress in Chemistry, 2025 , 37(9) : 1373 -1383 . DOI: 10.7536/PC20250108
表1 主要利用核能国家的压水堆单机组产生废物包年产生量的代表数据Table 1 Representative data of annual waste packages generated by a single unit PWR in the main countries that use nuclear energy |
| Country | USA | France | Japan | Spain | Belgium | Republic of South Korea |
|---|---|---|---|---|---|---|
| Median value of waste package generation per unit (m³) | 20 | 84 | 8 | 46 | 23 | 52 |
| Optimal level of waste package generation per unit (m³) | 7 | 45 | 6 | 30 | 21 | 11 |
表2 核电厂已整备放射性废物存量清单Table 2 Inventory list of prepared radioactive waste of a nuclear power plant |
| No. | Nuclear Power Plant | Waste volume (m³) | |||||
|---|---|---|---|---|---|---|---|
| Evaporative residue | Spent resin | Sediment | Water filter | Technical waste | Total | ||
| 1 | Qinshan NPP | 1371.6 | 0.0 | 0.0 | 99.4 | 778.9 | 2249.9 |
| 2 | Qinshan No.2 NPP | 394.4 | 466.8 | 0.0 | 128.9 | 2342.0 | 3332.1 |
| 3 | Qinshan No.3 NPP | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| 4 | Dayawan NPP | 108.0 | 370.0 | 14.0 | 83.7 | 1441.8 | 2017.5 |
| 5 | Ling'ao NPP (Phase I) | 106.0 | 416.0 | 4.0 | 138.1 | 618.6 | 1282.7 |
| 6 | Ling'ao NPP (Phase II) | 70.4 | 122.8 | 0.0 | 38.4 | 346.0 | 577.6 |
| 7 | Jiangsu Tianwan NPP | 475.4 | 1086.4 | 0.0 | 0.0 | 653.6 | 2215.4 |
| 8 | Hongyanhe NPP | 24.8 | 97.2 | 0.0 | 73.2 | 726.0 | 921.2 |
| 9 | Ningde NPP | 67.2 | 90.8 | 0.0 | 66.4 | 426.0 | 650.4 |
| 10 | Yangjiang NPP | 72.8 | 69.6 | 0.0 | 27.4 | 65.2 | 235.0 |
| 11 | Fuqing NPP | 102.4 | 77.2 | 8.8 | 0.0 | 457.6 | 646 |
| 12 | Fangjiashan NPP | 66.4 | 33.6 | 0.0 | 17.4 | 206.6 | 324 |
| 13 | Changjiang NPP | 36.0 | 20.8 | 0.0 | 2.0 | 112.0 | 170.8 |
| 14 | Fangchenggang NPP | 8.0 | 40.4 | 0.0 | 14.8 | 2.4 | 65.6 |
| 15 | Haiyang NPP | 0.0 | 0.0 | 0.0 | 0.0 | 1.0 | 1.0 |
| 16 | Taishan NPP | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| 17 | Sanmen NPP | 0.0 | 0.0 | 0.0 | 0.1 | 0.0 | 0.1 |
| Total | 2903.4 | 2891.6 | 26.8 | 689.8 | 8177.7 | 14 689.3 | |
表3 放射性废水情况统计Table 3 Statistics of radioactive wastewater |
| Type | Characteristics | Radioactivity | Main Source | Estimated Wastewater Volume per Unit per Year |
|---|---|---|---|---|
| Process Wastewater | Low conductivity, good water quality, few impurities | Potentially high radioactive activity | Discharged and flushing water, or leakage from the primary loop, flushing and drainage from the desalter | 2250 m3/a |
| Chemical Wastewater | High conductivity, poor water quality, and many impurities | Potentially high radioactive activity | Drainage from chemical cleaning and decontamination, and drainage from the radioactive analysis laboratory | 1500 m3/a |
| Ground Drainage | Poor water quality, many impurities | Low radioactive activity | Equipment drainage and leakage are collected within the radioactive control area | 6250 m³/a (including 1250 m³/a of service drainage) |
| Detergent-Containing Drainage | Poor water quality, many impurities | Extremely low radioactive activity | Decontamination water at the sanitation entrance and exit, drainage from the laundry for contaminated suits | 4000 m3/a |
| Conventional Nuclear Island Drainage | Few impurities | Radioactive activity possibly exceeds discharge limits | Discharge water from the second loop system in the turbine building, effluent of the secondary loop water purification system | - |
图2 核电厂典型固体废物处理工艺流程Fig.2 Typical solid waste treatment process flow of a nuclear power plant |
表4 部分电厂废液和固废处理方式Table 4 Waste liquid and solid waste treatment methods of some power plants |
| No. | Nuclear Power Plant | Waste Liquid Treatment | Spent Resin Treatment | Concentrate Treatment | Filter Core Treatment | Dry Miscellaneous Waste Treatment |
|---|---|---|---|---|---|---|
| 1 | Qinshan NPP | Evaporation, Ion Exchange | Temporary Storage | Cement Solidification | Overpressure + Cement Fixation | Pre-pressure and Barreling |
| 2 | Qinshan No.2 NPP | Filtration, Evaporation, Ion Exchange | Cement Solidification | Cement Solidification | Overpressure + Cement Fixation | Pre-pressure and Barreling |
| 3 | Qinshan No.3 NPP | Filtration, Ion Exchange | Temporary Storage | Cement Solidification | Cement Fixation | Pre-pressure and Barreling |
| 4 | Dayawan NPP | Filtration, Evaporation, Ion Exchange | Cement Solidification | Cement Solidification | Overpressure + Cement Fixation | Pre-pressure and Barreling |
| 5 | Ling'ao NPP | Filtration, Evaporation, Ion Exchange | Cement Solidification | Cement Solidification | Overpressure + Cement Fixation | Pre-pressure and Barreling |
| 6 | Jiangsu Tianwan NPP | Filtration, Evaporation, Ion Exchange | Hot State Overpressure | Evaporation and Compaction | Overpressure + Cement Fixation | Pre-pressure and Barreling |
| 7 | Yangjiang NPP | Ion Exchange + Evaporation, RO Membrane + Ion Exchange | Cement Solidification, Dehydration + HIC | Cement Solidification, Dehydration + HIC | Overpressure + Cement Fixation, Dehydration + HIC | Pre-pressure and Barreling |
| 8 | Fuqin NPP | Filtration, Evaporation, Ion Exchange | Cement Solidification | Cement Solidification | Overpressure + Cement Fixation | Temporary Bagging |
| 9 | Fangjiashan NPP | Filtration, Evaporation, Ion Exchange | Cement Solidification | Cement Solidification | Overpressure + Cement Fixation | Pre-pressure and Barreling |
| 10 | Changjiang NPP | Filtration, Evaporation, Ion Exchange | Cement Solidification | Cement Solidification | Overpressure + Cement Fixation | Pre-pressure and Barreling |
| 11 | Sanmen NPP | RO Membrane + Ion Exchange | Hot State Overpressure | Cement Solidification | Overpressure + Cement Fixation | Temporary Bagging |
| 12 | Haiyang NPP | Filtration + Ion Exchange | Dehydration + HIC | Dehydration + HIC | Dehydration + HIC | Pre-pressure and Barreling |
表5 部分电厂平均预估固废产生量Table 5 Average estimated solid waste generation of some power plants |
| Nuclear Power Plant | Qinshan | Qinshan(Ⅱ) | Qinshan (Ⅲ) | Fangjiashan | Fuqing | Hainan | Tianwan | Sanmen | Average (m3/Reactor) |
|---|---|---|---|---|---|---|---|---|---|
| Number of Units | 1 | 4 | 2 | 2 | 4 | 2 | 4 | 2 | |
| Technical Waste | 32.8 | 141.8 | 56.8 | 37.4 | 121.1 | 24 | 54.6 | 28.1 | 23.6 |
| Air Filter | 1.2 | 24.4 | 2.7 | 8.7 | 11.4 | 0.6 | 0.9 | 0.7 | 2.4 |
| Water Filter | 0.7 | 3.9 | 12.2 | 1 | 3.1 | 0 | 0 | 0.7 | 1.0 |
| Spent Resin | 3.3 | 25.8 | 14 | 10.9 | 52.8 | 35.6 | 93.7 | 4.3 | 11.4 |
| Concentrated Liquid | 25.3 | 5.8 | 0 | 18 | 16 | 17 | 36 | 0.4 | 5.6 |
| Membrane Waste | -- | -- | -- | -- | -- | -- | -- | 0.2 | 0.0 |
| Molecular Sieve | -- | -- | 3.6 | -- | -- | -- | -- | -- | 0.2 |
| Total | 63.3 | 201.7 | 89.3 | 76 | 204.4 | 77.2 | 185.2 | 34.4 | 44.2 |
| Average (m3/ Reactor) | 63.3 | 50.4 | 44.7 | 38 | 51.1 | 38.6 | 46.3 | 17.2 |
表6 液体废物处理技术总结Table 6 Summary of liquid waste treatment technology |
| Treatment Method | Applicable Objects | Decontamination Factor | Characteristics | Main Application Situations |
|---|---|---|---|---|
| Filtration | Radioactive wastewater with uncertain chemical components and usually radioactive activity lower than the discharge limit | Relatively low | Usually used as a pretreatment | Almost all nuclear power plants |
| Ion Exchange | Radioactive wastewater with low chemical content and radioactive activity higher than the discharge limit | 10~100, the decontamination factor will increase to some extent when a multiple-level combination is used | Widely applied | Almost all pressurized water reactor nuclear power plants |
| Evaporation | Radioactive wastewater with high chemical content or complex components and radioactive activity higher than the discharge limit | 103~106 | High volume reduction, high cost for equipment, construction and operation | USA, Russia, France, Germany, Japan, China |
| Chemical Precipitation | Radioactive wastewater with low requirements for purification, and radioactive activity higher than the discharge limit | Up to104 | High requirements for radiation detection and reagent addition equipment | USA |
| Membrane filtration | Radioactive wastewater with low chemical content and radioactive activity higher than the discharge limit | 10~103 | Membranes need to be cleaned regularly | USA, Canada |
| [1] |
China Nuclear Energy Association.National nuclear power operation situation (January-December 2024). 2025
( 中国核能行业协会.全国核电运行情况(2024年1-12月). 2025).
|
| [2] |
( 高彦锋, 石西森, 吕钢. 中国核电, 2020, 13(6): 774).
|
| [3] |
( 吴浩, 徐春艳, 刘新华, 魏方欣. 核安全, 2013, 12(S1): 155).
|
| [4] |
Ministry of Environmental Protection of the People’s Republic of China, General Administration of Quality Supervision, Inspection and Quarantine of the People’s Republic of China. Technical requirements for discharge of radioactive liquid effluents from nuclear power plant (GB14587-2011). Beijing: China Environmental Science Press, 2011
环境保护部,国家质量监督检验检疫总局. 核电厂放射性液态流出物排放技术要求( GB 14587-2011). 北京: 中国环境科学出版社, 2011).
|
| [5] |
李永青, 陈勤, 薛明,彭小红. 中国环境科学学会2009年学术年会论文集
( 第二卷),中国环境科学学会2009年学术年会. 北京: 北京航空航天大学出版社, 2009, 46 ) .
|
| [6] |
( 孙寿华, 冉洺东, 林力, 刘文磊, 李振臣, 李文钰. 核动力工程, 2019, 40(6): 1).
|
| [7] |
( 邵延江, 李莉, 穆建波, 赵大鹏, 龙泊康, 车建业. 辐射防护, 2020, 40(2): 137).
|
| [8] |
( 李明, 马兴均, 陈莉, 高峰, 李炳林, 沙沙. 核动力工程, 2018, 39(2): 37).
|
| [9] |
( 过灵飞, 何正忠, 肖德涛, 黎世铖, 林姝婧. 核化学与放射化学, 2021, 43(2): 163).
|
| [10] |
( 张玮钰, 金畅, 肖德涛. 核化学与放射化学, 2017, 39(2): 183).
|
| [11] |
( 金畅, 喻翠云, 肖德涛, 冯旭, 李超. 核化学与放射化学, 2015, 37(1): 45).
|
| [12] |
张志银, 严沧生, 黄来喜. 核电厂放射性废物最小化. 北京: 中国原子能出版社, 2013).
|
| [13] |
|
| [14] |
( 方祥洪, 马若霞, 杨彬. 山东化工, 2016, 45(16): 197).
|
| [15] |
|
| [16] |
|
| [17] |
( 魏新渝, 马鸿宾, 熊小伟, 王一川, 谭承军, 方圆, 王志. 水处理技术, 2015, 41(12): 10).
|
| [18] |
( 刘红娟, 吴仁杰, 谢水波, 刘迎九. 材料工程, 2019, 47(10): 22).
|
| [19] |
|
| [20] |
( 于静, 王建龙. 核化学与放射化学, 2018, 40(2): 81).
|
| [21] |
( 于静, 王建龙, 蒋翼周. 清华大学学报(自然科学版), 2016, 56(3): 312).
|
| [22] |
刘传耀. 华东师范大学博士论文, 2021).
|
| [23] |
( 周义朋, 王晓伟, 贾铭椿, 杜志辉, 梁成强. 现代化工, 2022, 42(2): 117).
|
| [24] |
( 谢林蓓, 孟笑鹏. 化学工程与装备, 2024, (12): 173).
|
| [25] |
( 洪业, 张华, 王浩. 同位素, 2022, 35(4): 324).
|
| [26] |
刘舒雅. 华北电力大学硕士论文硕士论文, 2024).
|
| [27] |
( 张安睿, 艾玥洁. 化学进展, 2020, 32(10): 1564).
|
| [28] |
( 张慧迪, 李子杰, 石伟群. 化学进展, 2023, 35(3): 475).
|
| [29] |
|
| [30] |
( 黄鑫水, 张宁, 郭涵, 孙玉兵. 中国科学(化学), 2020, 50(8): 882).
|
| [31] |
|
| [32] |
|
| [33] |
( 李鑫, 胡洪营, 余骏一, 赵文玉. 环境科学, 2016, 37(5): 1858).
|
| [34] |
( 姜筝, 丰俊东. 水处理技术, 2014, 40(9): 34).
|
| [35] |
|
| [36] |
|
| [37] |
|
| [38] |
任萌, 刘小龙, 康武, 李佳. 《环境工程》2019年全国学术年会论文集, 《环境工程》2019年全国学术年会. 北京: 《环境工程》编委会、 工业建筑杂志社有限公司, 2019).
|
| [39] |
|
| [40] |
( 鲁芸芸, 曹其如, 陈云明, 黄立娟, 白晓峰, 李兵, 冯亮. 核动力工程, 2013, 34(6): 161).
|
| [41] |
|
| [42] |
|
| [43] |
Safety regulations for near surface disposal of low and medium level radioactive solid waste( GB 9132-2018). Beijing: Ministry of Ecology and Environment, State Administration for Market Regulation, 2018
生态环境部, 国家市场监督管理总局. 低、 中水平放射性固体废物近地表处置安全规定( GB 9132-2018). 北京: 中国环境科学出版社, 2018).
|
| [44] |
( 蒋磊, 王昭, 罗劲松. 辐射防护通讯, 2020, 40(2): 35).
|
| [45] |
( 赵亚珂. 科技视界, 2020, (2): 188).
|
| [46] |
( 余达万, 徐宏明, 周辰昊, 郭喜良, 余达宇, 姜春辉. 辐射防护, 2019, 39(3): 213).
|
| [47] |
( 李琦, 苟全录, 余小东. 辐射防护, 2018, 38(1): 80).
|
| [48] |
( 刘铁军, 孙学强. 辐射防护, 2018, 38(1): 58).
|
| [49] |
( 逯馨华, 张红见, 魏方欣, 崔聪, 刘婷. 核安全, 2017, 16(3): 55).
|
| [50] |
( 陆科峰, 钟香斌, 李超, 张鹏, 卢忠诚. 辐射防护, 2015, 35(4): 243).
|
/
| 〈 |
|
〉 |