

Research Progress of High Temperature CO2 Corrosion Inhibitors
Received date: 2025-04-21
Revised date: 2025-07-08
Online published: 2025-10-15
Supported by
National Natural Science Foundation of China(22378309)
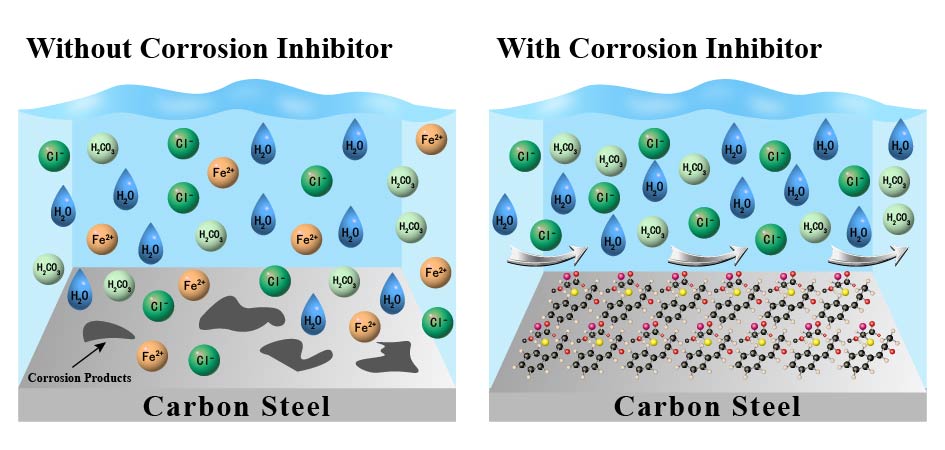
Carbon dioxide corrosion is one of the most common corrosion types of steel materials in the exploitation of oil and gas fields. Surprisingly, the corrosion caused by carbonic acid at the same pH is even more severe than hydrochloric acid, which has become an important factor limiting the development of the oil and gas industry. The use of corrosion inhibitors is the most economical and effective method to control CO2 corrosion. With the development of oil and gas drilling operations to deeper wells, CO2 corrosion under high temperature and pressure environment becomes increasingly prominent. This paper introduces the mechanism of high-temperature CO2 corrosion of carbon steel. The paper reviews the domestic and foreign research on corrosion inhibitors for high temperature CO2 and high S environment, mainly including imidazolines, quaternary ammonium salts, natural extracts and other corrosion inhibitors and analyzes corresponding characteristics. Finally, the future development direction of high temperature CO2 inhibitor was prospected.
1 Introduction
2 Corrosion mechanism of carbon dioxide on carbon steel at high temperature
3 Types of high-temperature CO2 corrosion inhibitors
3.1 Imidazoline high-temperature corrosion inhibitor
3.2 Quaternary ammonium salt high-temperature corrosion inhibitor
3.3 Natural extract high-temperature corrosion inhibitor
4 High temperature corrosion inhibitor in high S environment
5 Conclusion and outlook
Wenying Yuan , Shuang Jiang , Xiaoou Zhang , Tianyong Zhang , Ruitao Wang , Huaiyuan Wang . Research Progress of High Temperature CO2 Corrosion Inhibitors[J]. Progress in Chemistry, 2025 , 37(10) : 1428 -1437 . DOI: 10.7536/PC20250413
| [1] |
|
| [2] |
(顾林, 丁纪恒, 余海斌. 化学进展, 2016, 28(5): 737).
|
| [3] |
(侯保荣, 徐滨士, 徐惠彬. 中国腐蚀成本. 北京: 科学出版社, 2017).
|
| [4] |
|
| [5] |
(杨学耕, 陈慎豪, 马厚义, 全贞兰, 李德刚. 化学进展, 2003, 15(2): 123).
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
|
| [19] |
(韩敏娜, 于洪江, 周建猛. 广州化工, 2016, 44(19): 159).
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
(唐泽玮, 慕立俊, 周志平, 黄伟, 范希良, 周佩, 何淼, 李明星. 油田化学, 2019, 36(3): 472).
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
(尹成先, 阮林华, 丁万成, 兰新哲, 冯耀荣, 白真权. 材料保护, 2006, 39(2): 1).
|
| [30] |
(李继勇, 赵俊桥, 邵红云, 张云芝, 董洁, 夏凌燕, 胡松青. 油田化学, 2021, 38(1): 152).
|
| [31] |
(刘冬梅. 西安石油大学学报(自然科学版), 2020, 35(6): 65).
|
| [32] |
(王虎, 肖博文, 许家科, 谢娟, 崔海龙, 段明. 石油化工腐蚀与防护, 2020, 37(1): 8).
|
| [33] |
(刘寒梅, 张通, 宋建涛. 当代化工, 2022, 51(3): 585).
|
| [34] |
|
| [35] |
(张宇, 郭继香, 杨矞琦. 精细化工, 2019, 36(11): 2309).
|
| [36] |
|
| [37] |
|
| [38] |
|
| [39] |
|
| [40] |
|
| [41] |
(杨明地, 孙琳钧, 蒋立宏. 中国石油和化工标准与质量, 2013, 33(11): 37).
|
| [42] |
(曾文广, 李芳, 胡广强. 应用化工, 2020, 49(2): 57).
|
| [43] |
(曾文广, 李芳, 胡广强, 郭玉洁, 张江江, 刘斌. 安全、健康和环境, 2020, 20(10): 34).
|
| [44] |
|
| [45] |
(姜为民. 石油工程建设, 2021, 47(4): 34).
|
| [46] |
|
| [47] |
|
| [48] |
|
/
| 〈 |
|
〉 |