

Research Progress of Durable Slippery Liquid Infused Porous Surface
Received date: 2025-03-27
Revised date: 2025-06-09
Online published: 2025-10-15
Supported by
Shenzhen Municipal Science and Technology Research Program(CJGJZD20230724092159002)
National Natural Science Foundation of China(22378253)
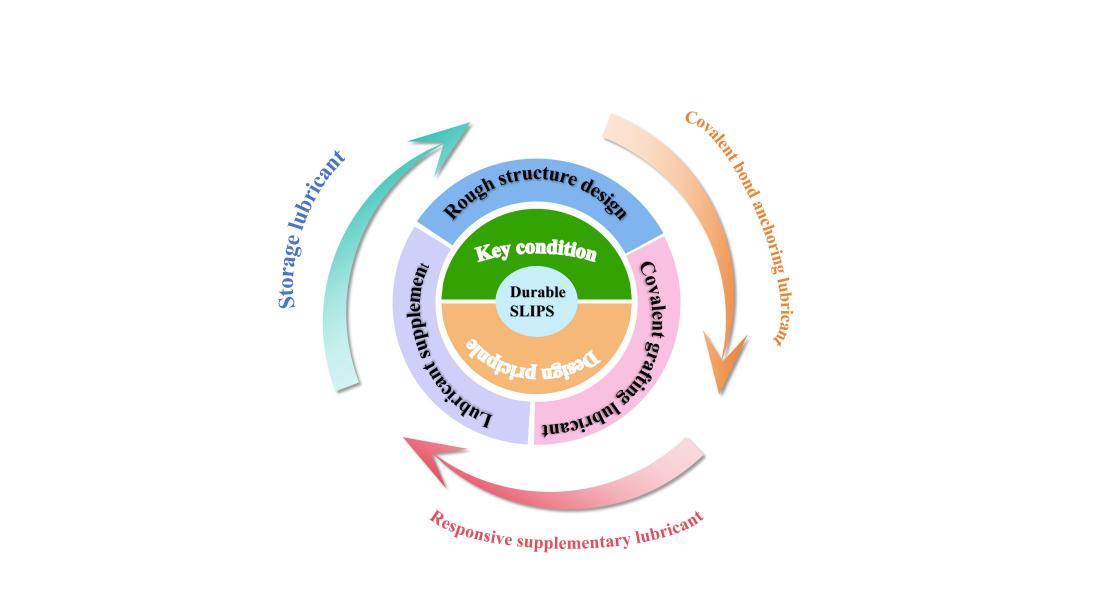
The slippery liquid-infused porous surface (SLIPS), owing to its unique liquid-repellent properties, has been widely applied in diverse fields such as anti-fouling, anti-corrosion, de-icing and droplet manipulation. However, the SLIPS tend to experience lubricant depletion when subjected to external mechanical abrasion, consequently diminishing or even completely losing their liquid-repellent properties. In light of this, this paper begins by exploring the three foundational principles of SLIPS design, and clarifies the guiding role these theories in the design process. Five critical requirements for fabricating durable SLIPS are also systematically summarized. Furthermore, by integrating global research progress, three strategies to enhance the durability of SLIPS are distilled. These strategies involve optimizing the rough structure to improve mechanical stability, anchoring lubricants through covalent grafting techniques to ensure long-term lubrication, and establishing lubricant replenishment mechanisms to sustain the durability of lubricating layer. A concise evaluation of their respective advantages and limitations is also provided. Finally, based on the bottlenecks of these strategies, key challenges in improving the mechanical durability of SLIPS are identified. Then, the future research directions are proposed, including optimizing nano-rough substrate design, expanding the functionalization of polymer molecular brushes, developing green and environmentally friendly lubricants, and enhancing SLIPS durability through multidimensional engineering approaches. In short, this paper aims to provide a new idea and way for the further study and application of SLIPS.
1 Introduction
2 Design principles for SLIPS
2.1 Spontaneous infiltration of lubricant into rough substrates
2.2 Complete wetting of rough substrate by lubricant
2.3 Thermodynamic immiscibility between lubricant and environmental liquid
3 Design and preparation of SLIPS
3.1 Rough structure design
3.2 Covalent grafting lubricant
3.3 Lubricant supplement
4 Conclusion and outlook
Ye Sun , Yan Bao , Ruyue Guo , Lu Gao , Chao Liu , Jingwei Yang . Research Progress of Durable Slippery Liquid Infused Porous Surface[J]. Progress in Chemistry, 2025 , 37(10) : 1513 -1524 . DOI: 10.7536/PC20250316
表1 3种不同耐久性提升策略的原理及优缺点比较Table 1 Compares the principles, advantages and disadvantages of three different durability improvement strategies |
| Strategy | Principle | Advantages | Disadvantages | |
|---|---|---|---|---|
| Rough structure design | Capillary force and van der Waals force | Wide compatibility | Environmental sensitivity | |
| Covalent grafting lubricant | Covalent bond strengthening force | Strong adhesion | Preparation complexity | |
| Lubricant supplement | Responsive lubricant | Responsive factors combined with lubricants | Wide applicability scenarios | Poor cyclic stability |
| Responsive substrate | Responsive factors for constructing rough substrates | Flexible design | Uncontrollable response speed | |
图5 (a) SLIPS制备示意图。通过水热法在微金字塔上生长ZnO纳米线,并通过光催化反应将PDMS电刷接枝到ZnO纳米线上;(b) 在KOH溶液中的蚀刻过程;(c) ZnO纳米线与PDMS接枝反应[52]Fig.5 (a) Schematic of the SLIPS preparation. The ZnO nanowires grew on micropyramids by the hydrothermal method and the PDMS brush grafted on ZnO nanowires by light-catalyzed reaction. (b) Etching process in KOH solutions. (c) The reaction of grafting in ZnO nanowires with PDMS[52] |
| [1] |
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
|
| [19] |
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
(李丹丹. 哈尔滨工程大学博士论文, 2024).
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
|
| [36] |
|
| [37] |
|
| [38] |
|
| [39] |
|
| [40] |
|
| [41] |
|
| [42] |
|
| [43] |
|
| [44] |
|
| [45] |
|
| [46] |
|
| [47] |
|
| [48] |
|
| [49] |
|
| [50] |
|
| [51] |
|
| [52] |
|
| [53] |
|
| [54] |
|
| [55] |
|
| [56] |
|
| [57] |
|
| [58] |
|
| [59] |
|
| [60] |
|
| [61] |
|
| [62] |
|
| [63] |
|
| [64] |
|
| [65] |
|
| [66] |
|
| [67] |
|
| [68] |
|
| [69] |
|
| [70] |
|
| [71] |
|
| [72] |
|
| [73] |
|
| [74] |
|
| [75] |
|
| [76] |
|
| [77] |
|
| [78] |
|
| [79] |
|
| [80] |
|
| [81] |
|
| [82] |
|
| [83] |
|
| [84] |
|
| [85] |
|
| [86] |
|
| [87] |
|
| [88] |
|
| [89] |
|
| [90] |
|
| [91] |
|
| [92] |
|
/
| 〈 |
|
〉 |