

Application of Carbon-based Nanozymes in Disease Treatment and Prevention
Received date: 2025-03-07
Revised date: 2025-05-01
Online published: 2025-10-15
Supported by
National Natural Science Foundation of China(22074023)
Medical Scientific Research Foundation of Guangdong Province, China(A2024469)
Scientific Research Project of Traditional Chinese Medicine Bureau of Guangdong Province, China(20251213)
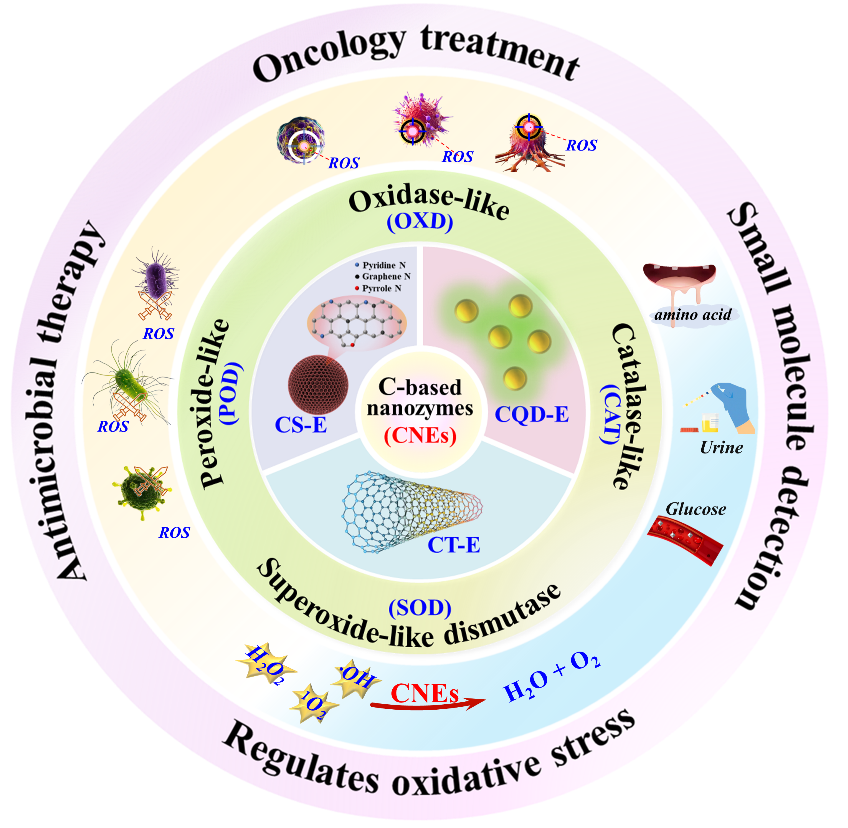
As a new generation of biomimetic enzyme catalytic materials, carbon-based nano-mimetic enzymes (CNEs) demonstrate significant value in the fields of sample analysis, environmental remediation and biomedicine, which is due to their multi-enzyme activity characteristics, such as peroxidase/oxidase. Compared with natural enzymes, CNEs exhibit advantages such as facile preparation, low cost, excellent stability, and, more importantly, the tailorable catalytic activity through structural modulation. These merits make them a promising next-generation alternative to the enzyme. Based on recent research progress over the past five years, this review summarizes the relationship between structural and catalytic activity from CNEs and elucidates the regulatory mechanism of their active site distribution and electronic structure on catalytic performance. It also discussed the strategies of heteroatom doping, secondary chemical modification, and environmental optimization factors for the influence on CNEs’ enzyme activity, and pointed out the effective enhancement approaches among them. Moreover, the application cases of enzyme-targeted therapy and preventive intervention in disease are reviewed. At the end of this paper, the future research prospect of new structural design and intelligent response system construction of CNEs is proposed, aiming to expand the application boundary of CNEs in the field of precision medicine and public health, and provide innovative ideas and strategies for solving global health problems. Finally, prospects for CNEs are proposed, focusing on novel structural designs and intelligent responsive system development. Hopefully, we can expand CNEs’ applications in precision medicine and public health domains, thereby providing innovative solutions for addressing global health challenges.
1 Introduction
2 Classification of structure and activity of carbon-based nanozymes
2.1 Carbon nano-spheres enzyme
2.2 Carbon nanotubes enzyme
2.3 Carbon quantum dot enzymes
2.4 Porous organic frameworks
2.5 Single-atom carbon-based nanozymes
2.6 Other
3 The influent factors the catalytic activity of carbon-based nanozymes
3.1 Heteroatom doping
3.2 Secondary chemical modification
3.3 Environmental condition control
4 Biomedicine application of carbon-based nanozymes
4.1 Small molecule screening
4.2 Regulates oxidative stress
4.3 Antimicrobial therapy
4.4 Oncology treatment
5 Conclusion and prospects
Meimei Liu , Shiyao Song , Zhihan Qu , Biyao Zhao , Runkun Zhang , Zhiming Huo . Application of Carbon-based Nanozymes in Disease Treatment and Prevention[J]. Progress in Chemistry, 2025 , 37(10) : 1540 -1554 . DOI: 10.7536/PC20250306
表1 CNEs的 类酶活性分类Table 1 Characteristics and classification of CNEs |
| Enzyme-like activity | Abbreviation | Catalytic mechanism | Relevant CNEs |
|---|---|---|---|
| Peroxidase | POD | Oxidize the substrate to oxidated products and H2O, by utilizing H2O2 as oxidant | CS-E[5,30-31]、CT-E[32-33]、CQDs-E[16,34-42]、SCNEs[18,29,43-44] |
| Oxidase | OXD | Oxidize the substrate in the presence of O2 | SCNEs[45]、CS-E[14,46-47]、CT-E[6,15]、CQDs-E[7] |
| Catalase | CAT | Decomposes H2O2 into water and oxygen | CS-E[48]、MOF[49]、SCNEs[29] |
| Superoxide dismutase | SOD | Catalyzes the generation of O2 and H2O2 by O2- | C-Dots[50]、CS-E[51]、Fullerene[52] |
| Catechol oxidase | - | Cu2+ coordinates with the -OH of catechols. The interconversion between Cu2+ and Cu+ catalyzes the oxidation of catechols | MOF[53-57] |
| Hydrolase | - | (1) Phosphatase: hydrolyzes the PO43- from organophosphorus pesticides to produce alcohols or phenolic compounds and phosphate ions (2) Esterases: hydrolyze esters to produce alcohols and carboxylic acids Others like uricase and aminoacylase share similar mechanisms | COF[58]、CS-E[59] |
| Photo-excited oxidase | - | Photoexcitation produces excited electrons and holes that react with dissolved oxygen to form ROS oxidation substrates | CQDs-E[60]、COF[61] |
图1 (a) 富氮CS-E的结构及位点示意图及TEM图[59]。(b) 经钝化的CT-E催化活性变化图及O-CNTs-7的TEM图[33]。(c) CQD-E 分类:GQDs-E和CPDs-E 。插图为His-GQD(例)的尺寸分布(左下)和HRTEM图(右上)[66]。(d) MOF和COF示意图,及MOF-818@PMOF(Fe)[53]的SEM图。(e) SCNEs的结构示意图及RhN4的TEM图[29]Fig. 1 (a) Schematic diagram and TEM diagram of the structure and loci of nitrogen-rich CS-E[59]. Copyright 2023, Springer Nature; (b) changes in catalytic activity of passivated CT-E and TEM of O-CNTs-7[33]. Copyright 2022 Elsevier; (c) the classification of CQD-E: GQDs-E and CPDs-E. The inset is the size distribution of His-GQD (bottom left) and the HRTEM plot (top right)[66]. Copyright 2020 Springer Nature; (d) schematic diagram of MOF and COF, and SEM plot of MOF-818@PMOF(Fe)[53]. Copyright 2023 American Chemical Society; (e) schematic diagram of the structure of SCNEs and TEM diagram of RhN4[29]. Copyright 2022 Springer Nature |
图3 杂原子掺杂和二次化学改性对纳米酶种类和活性的影响。(a) 具类POD活性的氮掺杂CNEs。(b) 具类SOD活性的过渡金属(如Ce、Cu、Co等)掺杂CNEs。(c) 具类POD活性的金属-N元素共掺杂CNEs。(d) CNEs的二次化学改性:功能化修饰及刻蚀技术Fig.3 Effects of heteroatom doping and secondary chemical modification on the types and activities of nanozymes. (a)N-doped nanomaterials exhibit peroxidase-like activity. (b) Transition metals (e.g., Ce, Cu, Co, etc.) doped nanomaterials exhibit SOD-like enzyme activity. (c) Some metals co-doped with N elements exhibit peroxidase-like activity. (d) Secondary chemical modification of CNEs: functionalization and etching techniques. |
表2 CNEs在生物医学中应用及机制Table 2 Biomedical Applications and Mechanistic of CNEs |
| Classification | Mechanism | Mechanism diagrams | Ref |
|---|---|---|---|
| Small molecule screening (4.1) | In the presence of CNEs, substrate is oxidized to form a colored product (e.g., TMB → TMBox), which can be quenched quantitatively by the analytes. | 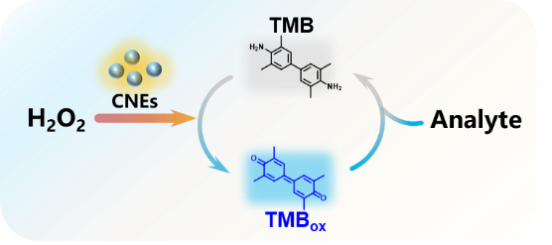 | 9,109~110 |
| Regulates oxidative stress (4.2) | CNEs quench free radicals such as ·O2-, H2O2 and •OH produced by metabolism to maintain oxidative-antioxidant balance. | 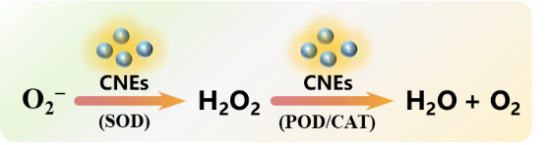 | 31,107,114~115 |
| Antimicrobial therapy (4.3) | CNEs catalyze the decomposition of oxidants to produce ROS, which destroys bacterial cell structure. | 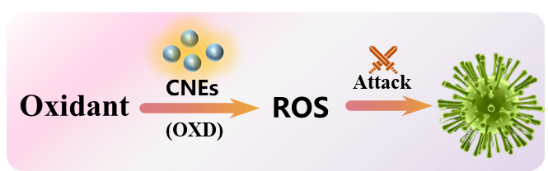 | 32,36,50,106,111 |
| Oncology treatment (4.4) | CNEs specifically bind to the FA receptors on the surface of the tumor. Oxidant was catalyzed and transformed to ROS, which activates the immune system and promotes the infiltration of immune cells into tumors, leading to the apoptosis of tumor cells. | 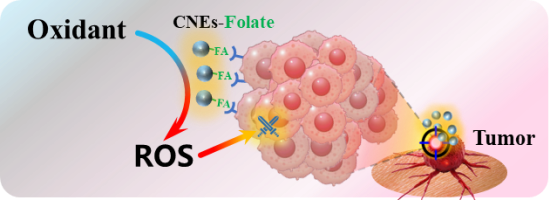 | 7,11,30,6,86 |
图4 (a) 含有不同浓度D-Pro的反应体系的吸收光谱[109]。(b) 具有抗ROS清除能力的CDs纳米酶用于抗感染治疗的示意图[114]。(c) 氧化酶样活性的FeSA-OLC应用于抗菌治疗[119]。(d) 通过ChA CQDs将免疫冷肿瘤转化为热肿瘤的拟议机制示意图[7]Fig.4 (a) Schematic diagram and absorption spectra of enzyme reaction systems on detecting D-Pro[109], Copyright 2022, Elsevier. (b) Schematic illustration of CDs nanozyme with scavenging ability against ROS for anti-inflammatory therapy[114], Copyright 2022, Frontiers Media S.A. (c) Oxidase-like activity of FeSA-OLC used for antimicrobial therapy[119], Copyright 2025 Elsevier. (d) Schematic diagram of the proposed mechanism for converting immunologically cold into hot tumors by ChA CQDs[7], Copyright 2022, American Chemical Society. |
| [1] |
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
|
| [19] |
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
|
| [36] |
|
| [37] |
|
| [38] |
|
| [39] |
|
| [40] |
|
| [41] |
|
| [42] |
|
| [43] |
|
| [44] |
|
| [45] |
|
| [46] |
|
| [47] |
|
| [48] |
|
| [49] |
|
| [50] |
|
| [51] |
|
| [52] |
|
| [53] |
|
| [54] |
|
| [55] |
|
| [56] |
|
| [57] |
|
| [58] |
|
| [59] |
|
| [60] |
|
| [61] |
|
| [62] |
|
| [63] |
|
| [64] |
|
| [65] |
|
| [66] |
|
| [67] |
|
| [68] |
|
| [69] |
|
| [70] |
|
| [71] |
|
| [72] |
|
| [73] |
|
| [74] |
|
| [75] |
|
| [76] |
|
| [77] |
|
| [78] |
|
| [79] |
|
| [80] |
|
| [81] |
|
| [82] |
|
| [83] |
|
| [84] |
|
| [85] |
|
| [86] |
|
| [87] |
|
| [88] |
|
| [89] |
|
| [90] |
|
| [91] |
|
| [92] |
|
| [93] |
|
| [94] |
|
| [95] |
|
| [96] |
|
| [97] |
|
| [98] |
|
| [99] |
|
| [100] |
|
| [101] |
|
| [102] |
|
| [103] |
|
| [104] |
|
| [105] |
|
| [106] |
|
| [107] |
|
| [108] |
|
| [109] |
|
| [110] |
|
| [111] |
|
| [112] |
|
| [113] |
|
| [114] |
|
| [115] |
|
| [116] |
|
| [117] |
|
| [118] |
|
| [119] |
|
| [120] |
|
/
| 〈 |
|
〉 |