

Progress of Electrodialysis in the Comprehensive Utilization of Brine Resources
Received date: 2025-02-24
Revised date: 2025-04-08
Online published: 2025-09-05
Supported by
The Science and Technology Major Projects of Xinjiang Autonomous Region(2022A03009-4)
The Science and Technology Key Project of Wuhan(2023010302020030)
The "TianchiTalent" Introduction Plan of the Xinjiang Uygur Autonomous Region
Brine resources are widely present in salt lakes, groundwater, and seawater. They are rich in many valuable elements such as lithium, potassium, magnesium, and boron, and thus possess significant economic value. With the rapid development of the new energy industry, especially the sharp increase in the demand for lithium resources, the comprehensive utilization of brine resources has become crucial for ensuring the sustainable supply of resources and promoting green development. However, traditional brine treatment methods, such as evaporation crystallization and chemical precipitation, have problems like high energy consumption, low separation precision, and environmental pollution. There is an urgent need for more efficient and environmentally friendly technical means. As a separation technology based on ion exchange membranes and the action of an electric field, electrodialysis technology has remarkable advantages such as high efficiency, energy conservation, and environmental friendliness, and has gradually become an important technology in brine resource treatment. This article introduces the principles of electrodialysis technology, including the working mechanisms of anion and cation membranes and bipolar membranes. By combining application cases, it explores the research progress of electrodialysis technology in the comprehensive utilization of brine resources. In terms of separation and extraction, this technology has a remarkable effect on the separation and extraction of elements such as lithium, boron, and potassium. It has outstanding advantages, especially in the extraction of lithium from brine with a high magnesium - to - lithium ratio. In the concentration process, it can achieve brine concentration with low energy consumption. In product processing, it can improve product purity and optimize the production process. Although electrodialysis technology has achieved remarkable results in the laboratory and pilot - scale stages, it still faces challenges such as the durability of membrane materials and equipment costs in large - scale industrial applications. In the future, electrodialysis technology is expected to develop synergistically with other technologies. Differentiated technical solutions will be developed according to the characteristics of different brine resources to achieve the full - component utilization of brine resources and promote the sustainable development of related industries.
1 Introduction
1.1 Brine resources
1.2 Comprehensive utilization of brine
2 Principles and application of electrodialysis
2.1 Principle of anion and cation membranes
2.2 Principle of bipolar membranes
2.3 Application case
3 Advances in the research and application of electrodialysis technology
3.1 Separation and extraction
3.2 Concentration
3.3 Product processing
4 Summary and outlook
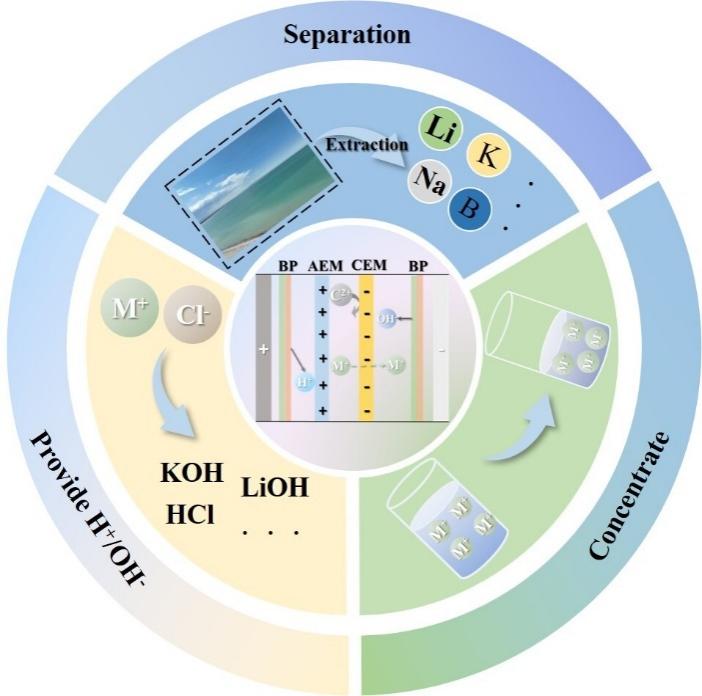
Key words: brine; electrodialysis; comprehensive utilization; ion exchange membrane
Liu Qianxin , Xia Kaisheng , Yang Zhen , Meng Yi , Tian Yunfeng , Chi Bo , Wu Yier , Liu Chenglin . Progress of Electrodialysis in the Comprehensive Utilization of Brine Resources[J]. Progress in Chemistry, 2025 , 37(9) : 1361 -1372 . DOI: 10.7536/PC20250209
表1 卤水综合利用各种方法的优缺点对比Table 1 Comparison of the advantages and disadvantages of various methods for comprehensive utilization of brine |
| Method | Advantages | Disadvantages | Ref |
|---|---|---|---|
| Evaporation Crystallization | 1.Simple operation 2.Low cost 3.Suitable for large-scale production | 1.High energy consumption. 2.Low separation precision. 3.Environmental risks. | 23-25 |
| Chemical Precipitation | 1.Fast reaction 2. Simple equipment 3. Low cost | 1.Chemical pollution. 2. Low selectivity for complex components. | 26-29 |
| Solvent Extraction | 1.High selectivity for rare elements (e.g., lithium) 2.Suitable for low-concentration brine | 1.High solvent cost and pollution risk. 2.Complex process | 30-31 |
| Ion Exchange | 1.High selectivity for specific ions (e.g., lithium, potassium) 2.Mild operating conditions | 1.High resin cost and fouling risk. 2.Limited capacity for large-scale use | 32-33 |
| Membrane Separation | 1.High efficiency and low energy consumption 2.Environmentally friendly 3.Suitable for multiple ions | 1.Membrane fouling and high maintenance cost. 2.High initial investment. | 34 |
| Electrodialysis | 1.High efficiency and low energy consumption 2.Environmentally friendly 3.Suitable for high Mg/Li ratio brine | 1.Limited to separating few elements. 2.Membrane fouling and high maintenance cost. | 35-36 |
表2 电渗析(ED)在卤水资源综合利用中的应用Table 2 Application of electrodialysis (ED) in the comprehensive utilization of brine resources |
| Case | Technology | Process | Outcomes | Ref |
|---|---|---|---|---|
| Lithium Extraction | ED + Adsorption | Pre-concentrate Li⁺ → Adsorption → LiOH | LiOH·H2O purity > 99.50% Energy consumption:4.15 kW·h/kg | 58-59 |
| selective electrodialysis (SED) | Separate Mg2⁺/Li⁺ | Li recovery:95.30% Energy consumption:0.391 kW·h/kg | 60 | |
| BMED + Precipitation | Pre-concentrate Li⁺ → Precipitation Li2CO3 → LiOH | LiOH purity > 98% Energy consumption:6.6 kW·h/kg | 61 | |
| BMED + NF + RO | NF → RO + CED → BMED → LiOH | Li recovery > 92% LiOH·H2O purity > 99.60% | 62 | |
| Potash Extraction | ED + Crystallization | Crystallization→ Separate K⁺/Na⁺ → KCl/NaCl | Mg2+/K+:7.11→0.92 K recovery > 74.83%, | 63 |
| SED | SED → Separate K⁺/Mg2⁺ → KCl | Mg2+/K+:10.31→1.15 K recovery > 89.06%, | 64 | |
| Boron Extraction | BMED | Separate B(OH)4- | B recovery :86.5% Energy consumption:31.5 kW·h/kg | 65 |
| Calcium and Magnesium Extraction | BMED + Crystallization | BMED + CO2 Crystallization → MgCO3 | Mg recovery:71.88% Energy consumption:1.22 kW·h/kg | 66 |
| Cylindrical Membrane ED | Cylindrical Membrane ED → Ca2+ | Ca recovery:30.17% | 67 |
| [1] |
|
| [2] |
|
| [3] |
|
| [4] |
( 陈欣怡, 夏开胜, 高强, 杨振, 李雨蝶, 孟伊, 陈亮, 刘成林. 化学进展, 2023, 35(10): 1519).
|
| [5] |
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
|
| [19] |
|
| [20] |
|
| [21] |
|
| [22] |
( 熊增华, 王兴富, 王石军, 童阳春, 吴盛斌. 盐湖研究, 2020, 8(4): 125).
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
( 储迎宇, 夏开胜, 高强, 杨振, 李雨蝶, 陈欣怡, 孟伊, 李珍, 俎波, 刘成林. 矿产保护与利用, 2023, 43(04): 130).
|
| [34] |
|
| [35] |
|
| [36] |
|
| [37] |
( 赵德军, 董玉林. 天津化工, 2024, 38(5):103).
|
| [38] |
( 李岩, 马珍, 宋兴福. 无机盐工业, 2023, 55(8): 84).
|
| [39] |
( 王石军, 靳玉倩, 杨晓东, 谢建明, 韩永福. 化工矿物与加工, 2024, 53(9): 34).
|
| [40] |
|
| [41] |
|
| [42] |
|
| [43] |
|
| [44] |
|
| [45] |
|
| [46] |
|
| [47] |
|
| [48] |
|
| [49] |
|
| [50] |
|
| [51] |
|
| [52] |
|
| [53] |
|
| [54] |
|
| [55] |
|
| [56] |
|
| [57] |
|
| [58] |
|
| [59] |
|
| [60] |
|
| [61] |
|
| [62] |
|
| [63] |
|
| [64] |
|
| [65] |
|
| [66] |
|
| [67] |
|
| [68] |
|
| [69] |
|
| [70] |
|
| [71] |
|
| [72] |
|
| [73] |
|
| [74] |
|
| [75] |
|
| [76] |
|
| [77] |
( 马珍. 无机盐工业, 2022, 54(10): 22).
|
| [78] |
|
| [79] |
|
| [80] |
|
| [81] |
|
| [82] |
|
| [83] |
|
| [84] |
|
| [85] |
|
| [86] |
|
| [87] |
|
| [88] |
|
| [89] |
|
| [90] |
|
| [91] |
|
| [92] |
|
| [93] |
|
| [94] |
|
| [95] |
|
| [96] |
|
| [97] |
|
| [98] |
|
| [99] |
|
| [100] |
|
/
| 〈 |
|
〉 |