

Synthesis and Application of Water-Soluble D-A-D Type Near Infrared Two-Region Fluorescent Molecules
Received date: 2025-02-24
Revised date: 2025-04-18
Online published: 2025-09-12
Supported by
National Natural Science Foundation of China(22375102)
National Natural Science Foundation of China(52373142)
National Natural Science Foundation of China(U24A2077)
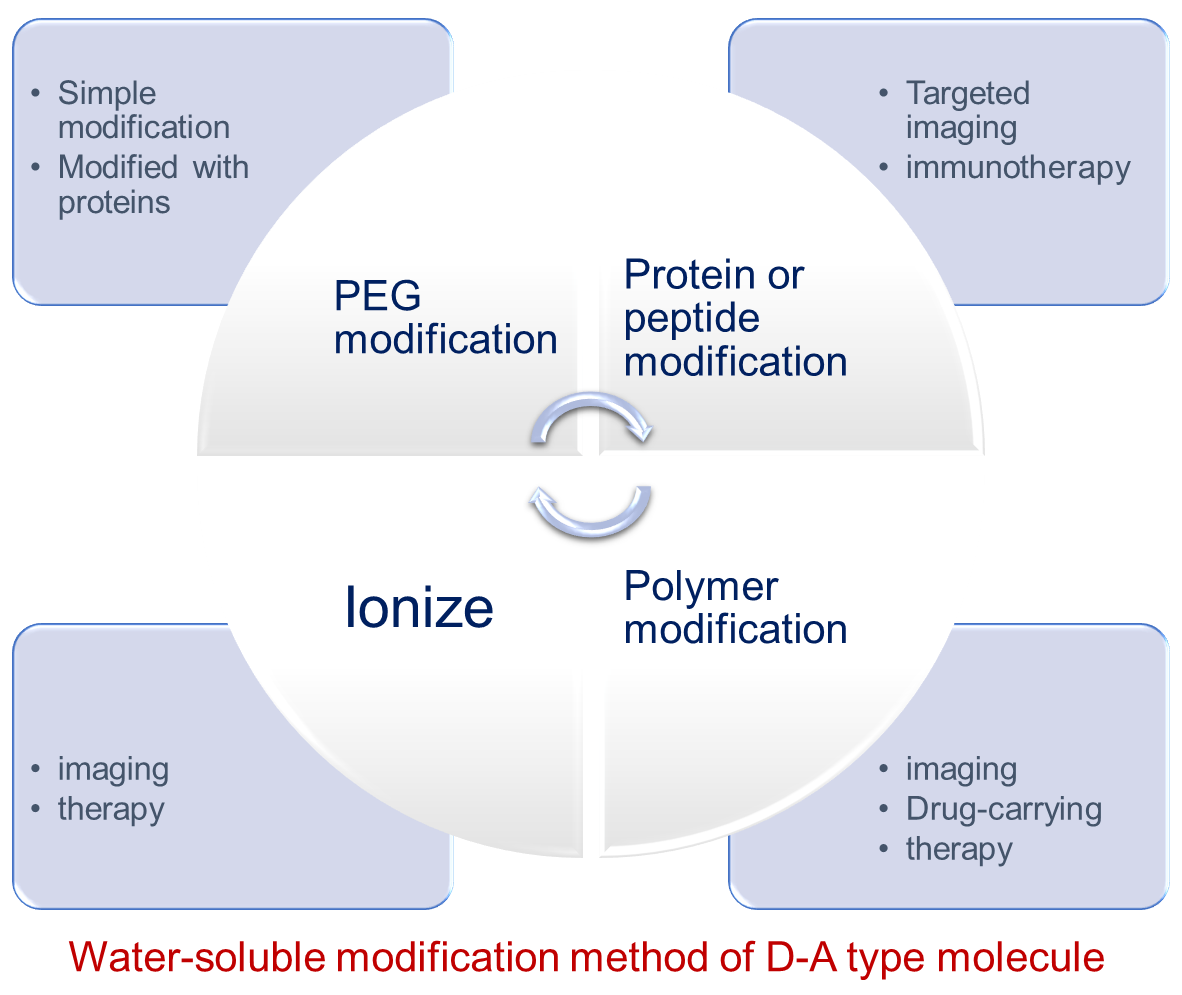
D-A-D molecule refers to a class of conjugated structure molecules composed of an electron donor and an electron acceptor. The NIR two-region fluorescence imaging dominated by such molecules has the advantages of good penetration effect and high imaging clarity. It has high application potential in clinical diagnosis. However, such molecules usually contain conjugated benzene ring structures. This means that the water solubility of these molecules is not good, which greatly limits the wider application of NIR-Ⅱregion fluorescence imaging. In recent years, D-A-D molecules have usually been modified to improve their water solubility. This review introduces four methods to improve water solubility by end-modified hydrophilic polyethylene glycol, modified other hydrophilic polymer chains, modified by protein or peptide, and end-ionized modification. The design methods and related applications of water-soluble D-A-D molecules are introduced in detail. Finally, the further development of water-soluble D-A-D small molecules in the field of NIR-Ⅱ region fluorescence imaging is prospect.
1 Introduction
2 PEG
2.1 Simple modified type
2.2 Targeted imaging
2.3 Drug-carrying
3 Polymer
3.1 Imaging
3.2 Drug-carrying and therapy
4 Protein, peptide
4.1 Targeted imaging
4.2 Therapy
5 Ionize
5.1 Imaging
5.2 Therapy
6 Conclusion and outlook
Yongzhen Chu , Pengfei Sun , Quli Fan . Synthesis and Application of Water-Soluble D-A-D Type Near Infrared Two-Region Fluorescent Molecules[J]. Progress in Chemistry, 2025 , 37(10) : 1555 -1568 . DOI: 10.7536/PC20250210
图6 (a) CDIR2肾脏清除率。 (b) TQFP-10肿瘤以及正常组织信号比。 (c) FBP912肾脏清除率。 (d) FBP912荧光成像。 (e) TQFP-10脑补肿瘤成像。 (f) CDIR2荧光成像[22-25]Fig.6 (a) CDIR2 renal clearance. (b) TQFP-10 tumor to normal tissue signal ratio. (c) FBP912 renal clearance. (d) FBP912 fluorescence imaging. (e) TQFP-10 imaging tumor imaging. (f) CDIR2 fluorescence imaging[22-25] |
| [1] |
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
|
| [19] |
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
(陈尚钰, 沈清明, 孙鹏飞, 范曲立. 高等学校化学学报, 2022, 43(12): 324).
|
| [27] |
(王超, 孙鹏飞, 范曲立, 高分子学报, 2022, (002): 053).
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
|
| [36] |
|
| [37] |
|
| [38] |
|
| [39] |
|
| [40] |
|
| [41] |
|
| [42] |
|
| [43] |
|
/
| 〈 |
|
〉 |