

Functional Construction and Application of Hemin-Based Mimetic Enzyme
Received date: 2025-05-12
Revised date: 2025-06-18
Online published: 2025-10-30
Supported by
China National Petroleum Corporation (CNPC) Critical Core Technology Tackling Project(2022GJ17)
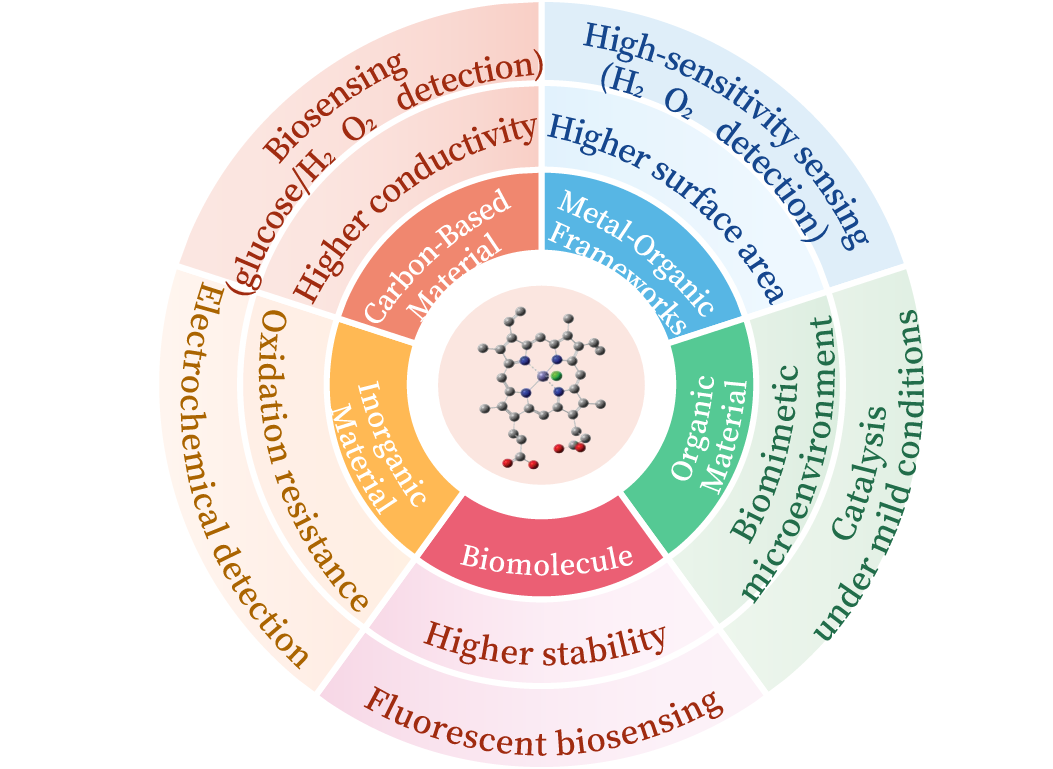
As a type of biomimetic catalyst, artificial enzymes can effectively overcome the limitations of natural enzymes in purification, storage, and recyclability. Hemin (Fe(Ⅲ)-protoporphyrin Ⅸ), serving as the essential cofactor in the active center of most peroxidases, possesses fundamental peroxidase-like catalytic activity due to its iron-porphyrin structure. However, native free hemin suffers from issues such as intermolecular self-aggregation, susceptibility to oxidative deactivation, and insufficient exposure of catalytic sites, leading to reduced catalytic efficiency and poor stability. Combining hemin with supporting materials to form hemin-based artificial enzymes can effectively inhibit hemin self-aggregation and oxidative degradation while simultaneously enhancing its catalytic activity and stability. This review primarily introduces several common types of hemin-based artificial enzymes. It summarizes and categorizes their construction and applications based on the underlying principles of the various support materials and the characteristics of the resulting hemin-based enzymes. Furthermore, it analyzes how the structural properties of different supports regulate the functions of the artificial enzymes and provides an outlook on their future development. Current challenges in designing and constructing hemin-based artificial enzymes include complex self-assembly processes and poor controllability during preparation. Future studies could focus on conducting in-depth physicochemical research on support materials to achieve a higher integration of hemin and support properties. This may involve establishing structure-activity relationship maps correlating the physicochemical properties of supports with the directional assembly of hemin molecules, implementing interface engineering strategies for synergistic optimization of hemin and carrier performance, or exploring alternative support materials with similar properties. The development of hemin-based artificial enzymes combining high catalytic activity with structural homogeneity is key to facilitating their practical applications across multiple fields.
Contents
1 Introduction
2 Hemin
3 Synthesis and application of hemin-based artificial enzymes
3.1 Carbon-supported hemin artificial enzymes
3.2 MOF-supported hemin artificial enzymes
3.3 Integration and applications of hemin-functionalized inorganic supports
3.4 Research and application of synthetic polymer-immobilized Hemin
3.5 Conjugation and applications of Hemin with biomacromolecules
4 Conclusion and outlook
Key words: hemin; artificial enzymes; metal-organic frameworks; biocatalysis
Ying Li , Lin Han , Tiantian Feng , Jian Li . Functional Construction and Application of Hemin-Based Mimetic Enzyme[J]. Progress in Chemistry, 2025 , 37(11) : 1652 -1660 . DOI: 10.7536/PC20250505
图1 血红素(Fe(Ⅲ)-前卟啉Ⅸ)的结构示意图:(a) 二维结构;(b) 三维结构俯视图;(c) 三维结构侧视图(为清晰起见,省略氢原子。其中灰色代表碳原子,红色代表氧原子,蓝色代表氮原子,绿色代表氯原子,蓝紫色代表铁原子[11]Fig.1 Schematic structure of hemin (Fe (Ⅲ)-protoporphyrin Ⅸ). (a) 2D structure; (b) top view 3D structure; (c) side view 3D structure; H atoms were omitted for clarity. Atoms are represented as spheres with the color coding: carbon (gray), oxygen (red), nitrogen (blue), chloride (green), and iron (blue violet)[11] |
表1 不同负载材料的物理化学特性对比Table 1 Comparison of physical and chemical properties of different load materials |
| Property | Graphene | Carbon Nanotubes | MOFs | BN | Supramolecular Hydrogel |
|---|---|---|---|---|---|
| SSA (m²/g) | High (2630) | Moderate-High (500) | Very High (>1000) | High (800) | Low (crosslinking) |
| Stability | High (inert) | High (thermal) | Moderate (hydrolysis-prone) | Very High (oxidation-resistant) | Moderate (environment-sensitive) |
| Biocompatibility | Moderate | Moderate | High (tunable) | Low | High |
| Functionalization | High (surface groups) | High (sidewall) | Very High (ligand-tunable) | Moderate | Moderate (self-assembly) |
| Anti-Dimerization | Strong | Strong | Very Strong (encapsulation) | Moderate | Strong (spatial) |
| [1] |
(何建川, 陈莲惠, 曹洪斌, 段烁. 化学研究与应用, 2021, 33(11): 2059).
|
| [2] |
|
| [3] |
|
| [4] |
(刘有芹, 颜芸, 沈含熙. 化学进展, 2005, 17(06): 1067).
|
| [5] |
(胡莹, 马桃林. 现代化工, 2018, 38(3): 71).
|
| [6] |
(王兰芝, 佘远斌, 徐未未, 张燕慧, 纪红兵. 化学进展, 2005, 17(4): 678).
|
| [7] |
(王慧悦, 胡欣, 胡玉静, 朱宁, 郭凯. 化学进展, 2022, 34(8): 1796).
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
(李庚, 申晓林, 孙新晓, 王佳, 袁其朋. 合成生物学, 2024, 5(6): 1498).
|
| [16] |
|
| [17] |
(赖欣宜, 王志勇, 郑永太, 陈永明. 化学进展, 2019, 31(6): 783).
|
| [18] |
|
| [19] |
|
| [20] |
(唐佳倩. 扬州大学硕士论文, 2016).
|
| [21] |
|
| [22] |
(杨春燕, 马云龙, 冯小琼, 张世英, 安长胜, 李劲风. 储能科学与技术, 2021, 10(2): 432).
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
(王桦, 张立燕, 李帅, 董敏敏, 冯路平. ZL 201610036442.0, 2018).
|
| [36] |
|
| [37] |
|
| [38] |
|
| [39] |
|
| [40] |
|
| [41] |
|
| [42] |
|
| [43] |
|
| [44] |
|
| [45] |
|
| [46] |
|
/
| 〈 |
|
〉 |