

Received date: 2025-04-07
Revised date: 2025-06-24
Online published: 2025-10-25
Supported by
National Natural Science Foundation of China(52272021)
National Natural Science Foundation of China(U23A20559)
National Natural Science Foundation of China(52232002)
Natural Science Foundation of Wuhan(2024040701010051)
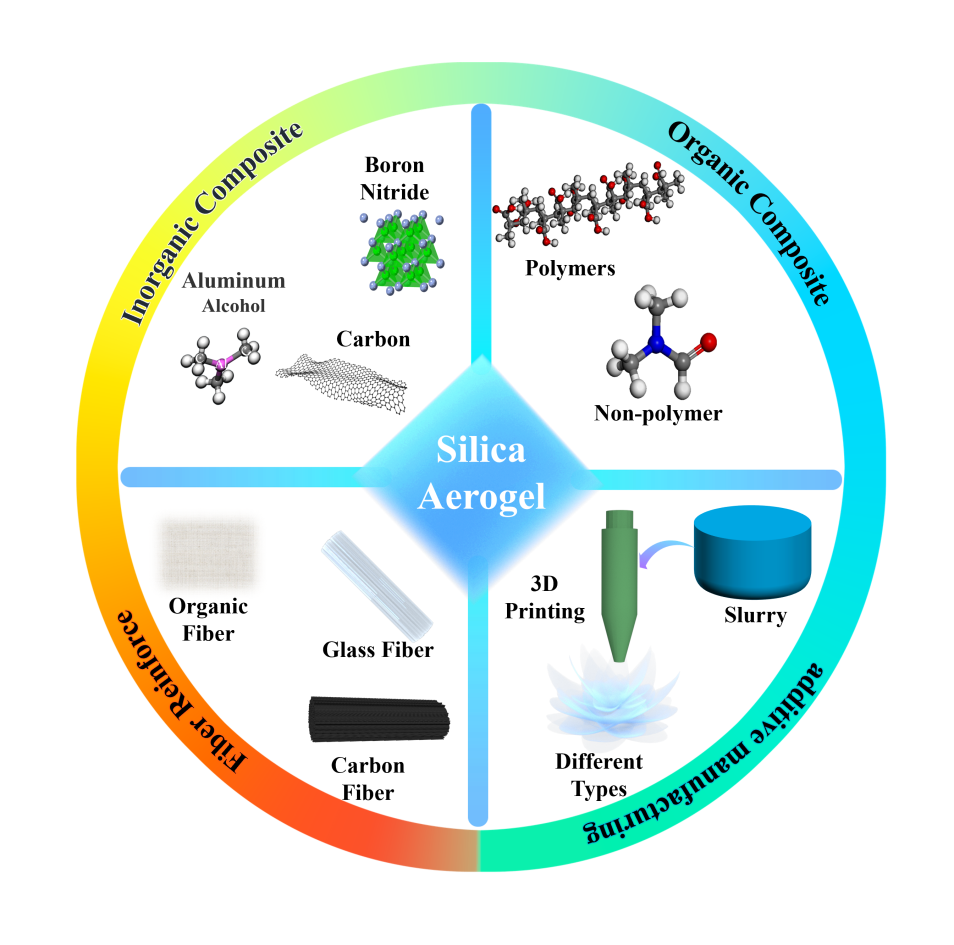
Silica composite aerogels, characterized by their extremely low density, high specific surface area, and remarkable porosity, have found extensive applications in high-temperature kilns, the oil and gas sector, aerospace, and various other advanced domains. Firstly, silica aerogels that have been composited through inorganic and organic compositing were thoroughly reviewed in this paper, as well as fiber reinforcement, including a comparative analysis of their thermal conductivity, compressive strength, porosity, density, and other significant physical properties. Secondly, the most recent strategies for additive manufacturing of silica composite aerogels are summarized. Finally, the challenges related to the fabrication and performance of silica composite aerogels and proposed future research directions for their advancement was addressed by this paper.
Contents
1 Introduction
2 Inorganic composite silica aerogel and preparation strategy
2.1 Aluminum oxide composite silica aerogel
2.2 Carbon composite silica aerogel
2.3 Non-oxide composite silica aerogel
3 Organic composite silica aerogel and preparation strategy
3.1 Polymer composite silica aerogel
3.2 Non-polymerzied composite silica aerogel
4 Fiber reinforced silica aerogel and preparation strategy
4.1 Carbonfiber
4.2 Glass fiber
4.3 Other inorganic fibers
4.4 Organic fiber
5 Additive manufacturing strategies for silica composite aerogels
6 Forntier application
6.1 Aerospace
6.2 Energy saving
6.3 Battery thermal management
7 Conculusion and outlook
Zichun Lin , Xinyue Wang , Qing Xu , Hongjuan Duan , Haijun Zhang . Silica Composite Aerogels[J]. Progress in Chemistry, 2025 , 37(11) : 1688 -1703 . DOI: 10.7536/PC20250406
表1 二氧化硅复合气凝胶的优缺点Table 1 Advantages and disadvantages of silica aerogel composite |
| Aerogel | Advantage | Disadvantage |
|---|---|---|
| Inorganic composite | High service temperature, structure stability | Limit service temperature, high brittleness |
| Organic composite | Complex heat transfer path, high-stiffness network | Carbonize, high deformation |
| Fiber reinforce | Shielding effect, distribute stress | Poor interface composite, increase heat transfer |
| 3D printing | Malleable, expand applications | High cost, low efficiency |
图2 无机物复合二氧化硅气凝胶性能增强的原理示意图Fig.2 Schematic representation of the principle of performance enhancement of inorganic composite silica aerogels |
表2 无机物复合二氧化硅气凝胶材料与性能对比a)Table 2 Comparison of inorganic composite silica aerogel materials and properties a) |
| Precursor | Composite Sources | Specific Surface area (m2/g) | Porosity (%) | Pore Value (cm3/g) | Density (g/cm3) | λ (W·m-1·K-1) | Compressive Strength (MPa)/Strain(%) | Contact Angel(°) | Ref | |
|---|---|---|---|---|---|---|---|---|---|---|
| TEOS | Aluminum Oxide | Aluminum sol | 616 | - | - | - | - | - | - | 20 |
| MTMS | 3-tert-Butylaluminum | - | - | - | 0.22 | 0.023 | - | - | 21 | |
| TEOS | Aluminum Oxide | Aluminum sol | 422 | 76.4 | 1.49 | 0.256 | 0.116 | - | - | 22 |
| TEOS | AlCl3 | - | - | - | - | 0.024 | - | - | 23 | |
| TEOS | ASB | 232 | 94.8 | 4.543 | 0.207 | 0.035 | 0.79/16.4 | - | 24 | |
| TEOS | Carbon | Graphene | 1096 | - | 2.67 | 0.105 | 0.018 | - | - | 30 |
| MTMS | Graphene oxide | - | - | - | - | 0.049 | 0.08/80.0 | - | 31 | |
| TEOS | CNT | 492 | 95.8 | 12.5 | 0.061 | 0.031 | - | - | 32 | |
| MTMS | CNT | 446.33 | - | 3.84 | 0.23 | 0.178 | 1.36/4.3 | - | 33 | |
| APTES | CNT | 463.4 | 95 | - | 0.613 | 0.031 | 35/65.6 | - | 34 | |
| MTMS, DEMS | Catechol, Formaldehyde | 774 | - | - | 0.118 | 0.044 | 2.09/44.2 | - | 35 | |
| TEOS | Particles | DMF, Ammonia solution | 566 | - | 1.12 | 2.25 | 0.04 | 20.3/15.0 | - | 39 |
| Octaphenylsilsesquioxane | Polyhedral Oligomeric Sesquicone | - | - | - | - | 0.028 | 26.4/82.6 | 158 | 40 | |
a) λ: Thermal conductivity; TEOS: ethyl orthosilicate; MTMS:methyltrimethoxysilane; TMCS: trimethylsilyl chloride; ASB: aluminum tri-sec-butoxide; DMF: N,N-dimethylformamide CNT: carbon nanotubes; APTES: (3-aminopropyl)triethoxysilane; DEMS: diethoxymethylsilane |
图6 有机复合二氧化硅气凝胶的增强机理示意图Fig.6 Schematic enhancement mechanism of organic composite silica aerogel |
表3 有机物复合二氧化硅气凝胶材料与性能对比a)Table 3 Comparison of organic composite silica aerogel materials and properties a) |
| Precursor | Composite sources | Specific surface area (m2/g) | Porosity (%) | Pore value (cm3/g) | Density (g/cm3) | λ (W·m-1·K-1) | Compressive Strength (MPa)/ Strain(%) | Contact angel (°) | Ref | |
|---|---|---|---|---|---|---|---|---|---|---|
| Silica aerogel powders | Polymers | 4,4'-Oxybisbenzenamine and 3,3',4,4'- Biphenyltetracarboxylic dianhydride | 609 | - | - | - | 0.018 | - | >150 | 41 |
| MPTES | PSA | - | 80 | - | 0.3 | 0.06 | >1.50/0.5 | - | 42 | |
| Silica aerogel powders | PVA | - | - | - | 0.13 | 0.019 | - | 150 | 43 | |
| TMOS | PEG | 303 | - | 0.19 | 0.093 | 0.023 | - | 136 | 44 | |
| Sodium Silicate | Methyl methacrylate | - | - | - | - | 0.019 | - | - | 45 | |
| Olivine | Polymers | cellulose | - | - | - | - | 0.027 | - | 135 | 46 |
| MTMS | cellulose | 958 | 98 | 0.72 | 0.055 | 0.023 | 0.09/80 | 140 | 47 | |
| Liquid Water Glass | Cellulose | 630 | - | - | - | 0.021 | 58.00/20 | - | 48 | |
| Liquid Water Glass | Bateria cellulose | 729 | - | 2.7 | - | - | - | 148 | 49 | |
| TEOS | PP | - | - | - | - | - | 2.02/70 | 145 | 50 | |
| TEOS, MTMS | Non-polymers | sorbital | 1193 | 92.3 | 2.28 | 0.15 | 0.041 | 0.21/10 | 151 | 55 |
| (3-glycidyloxypropyl)trimethoxysilane and (3-mercaptopropyl)trimethoxysilane, TEOS | Epoxy-thiol | 519 | 93.42 | - | 0.225 | 0.047 | 11.00/60 | - | 56 | |
| Liquid Water Glass | DMAC | 843 | - | 3.36 | - | - | - | - | 57 | |
a) MPTES: 3-mercaptopropyl triethoxysilane; PSA: polyarylacetylene; PVA: polyvinyl alcohol; PEG: polyethylene glycol; PP: polypropylene; BHNC: bifunctional hairy nanocellulose; APTES: (3-aminopropyl)triethoxysilane; TMOS: Tetramethyl orthosilicate; DMAC: N,N-dimethylacetamide |
图9 纤维复合二氧化硅气凝胶的增强机理示意图Fig.9 Schematic enhancement mechanism of fiber composite silica aerogel |
表4 纤维增强的二氧化硅气凝胶的性能对比a)Table 4 Comparison of properties of silica aerogels with fiber reinforced a) |
| Precursor | Composite sources | Specific surface area (m2/g) | Porosity (%) | Pore value (cm3/g) | Density (g/cm3) | λ (W·m-1·K-1) | Compressive strength (MPa)/Strain(%) | Contact angel (°) | Ref | |
|---|---|---|---|---|---|---|---|---|---|---|
| TEOS | Carbon Fiber | T700 polyacrylonitrile fiber | - | - | - | - | 0.112 | 17.01/40 | - | 60 |
| APTES | CF (hydroxyl modification) | 463.4 | 95 | - | 0.613 | 0.031 | 10.00/80 | - | 61 | |
| TEOS, TMOS | Glass fiber | 604 | 86 | 7.69 | 0.281 | 0.026 (664 ℃) | 0.75/55 | 150 | 64 | |
| Liquid Water Glass | - | - | - | 0.131 | 0.036 | - | 135 | 65 | ||
| TEOS | - | 95 | - | 0.104 | - | - | - | 66 | ||
| Slica Aerogel Particles | - | - | - | - | 0.022 (650 ℃) | - | - | 67 | ||
| TEOS | Other Fiber | Xonolite Fiber | - | - | 4.13 | 0.126 | 0.028 | - | - | 68 |
| MTES, TEOS | TiO2 nanotubes | - | - | - | - | 0.118 (1000 ℃) | - | - | 69 | |
| TEOS | Silica Nanowires | - | - | - | 0.11 | 0.039 | 1.38/60 | >140 | 70 | |
| TEOS | ZrO2-SiO2 fiber | 207.8 | 99.95 | - | - | 0.029 | - | - | 71 | |
| TEOS | Organic Fiber | Aramid pulp | 764 | - | 2.73 | 0.19 | 0.0261 | - | 145 | 72 |
| TEOS, VTMS | Aramid fibers | 973.3 | - | - | - | 0.022 | 0.35/60 | - | 73 | |
| TEOS | Kevlar pulp | 619 | 84.5 | - | 0.208 | 0.026 | 0.55/25 | 156 | 74 | |
| Liquid Water Glass | PP fibers | 644 | 87 | - | - | - | - | 120 | 75 | |
a) CF: carvon fiber MTES: methyltriethoxysilane; VTMS: vinyltrimethoxysilane; PP: polypropylene |
| [1] |
|
| [2] |
|
| [3] |
(颜富荣, 刘雅贤, 范龄元, 张梅, 郭敏. 材料工程 2023, 51(09): 1).
|
| [4] |
|
| [5] |
|
| [6] |
(贾光耀, 邓育新. 硅酸盐通报, 2004, 23(6): 91).
|
| [7] |
芦贻春, 李再耕. 耐火材料, 1995, (6): 326).
|
| [8] |
(何辉, 姜勇刚, 张忠明, 冯军宗, 李良军, 冯坚. 硅酸盐通报, 2022, 41(05): 1813).
|
| [9] |
(周长灵, 杨杰, 隋学叶, 刘瑞祥, 张文苑, 王重海. 现代技术陶瓷, 2014, 35(05): 11).
|
| [10] |
(杨景兴, 何凤梅, 于帆, 胡子君, 李俊宁. 宇航材料工艺, 2013, 43(02): 92).
|
| [11] |
(穆锐, 刘元雪, 刘晓英, 张育新, 姚未来, 任俊儒, 陈金锋, 成鑫磊, 杨秀明, 龚宏伟. 复合材料学报, 2024, 41(07): 3355).
|
| [12] |
|
| [13] |
|
| [14] |
(马利国, 孙艳荣, 李东来, 任富建, 李建平. 无机盐工业, 2020, 52(08): 11).
|
| [15] |
(范龄元, 张梅, 郭敏. 材料导报, 2022, 36(15): 5).
|
| [16] |
|
| [17] |
|
| [18] |
|
| [19] |
(商富强, 黄丽清, 李刚, 张宇, 蔡亚坤, 王慧敏, 董伟丽, 张磊, 刘悠. 材料导报. 2020, 34(10): 1).
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
(郭华超, 王良旺, 郭志强, 文芳, 何立粮, 熊磊. 化工新型材料. 2024, 52(9): 1).
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
|
| [36] |
|
| [37] |
|
| [38] |
|
| [39] |
|
| [40] |
|
| [41] |
|
| [42] |
|
| [43] |
|
| [44] |
|
| [45] |
|
| [46] |
|
| [47] |
|
| [48] |
|
| [49] |
|
| [50] |
|
| [51] |
|
| [52] |
|
| [53] |
|
| [54] |
(刘小妩, 李建梅, 段爱军. 石油科学通报, 2019, 4(3): 323).
|
| [55] |
|
| [56] |
|
| [57] |
|
| [58] |
|
| [59] |
|
| [60] |
|
| [61] |
|
| [62] |
|
| [63] |
|
| [64] |
|
| [65] |
|
| [66] |
|
| [67] |
|
| [68] |
|
| [69] |
|
| [70] |
|
| [71] |
|
| [72] |
|
| [73] |
|
| [74] |
|
| [75] |
|
| [76] |
|
| [77] |
|
| [78] |
|
| [79] |
|
| [80] |
|
| [81] |
|
| [82] |
|
| [83] |
|
| [84] |
|
| [85] |
|
| [86] |
|
| [87] |
|
/
| 〈 |
|
〉 |