

The Impact of Prenatal Perfluorinated and Polyfluoroalkyl Substances Exposure on Placental Structure and Function
Received date: 2025-06-12
Revised date: 2025-06-27
Online published: 2025-07-16
Supported by
National Natural Science Foundation of China(42322707)
National Natural Science Foundation of China(42325702)
National Natural Science Foundation of China(42407577)
Guangdong Provincial Key Laboratory of Soil and Groundwater Pollution Control(2023A1515140118)
Guangdong Basic and Applied Basic Research Foundation(2023B1212060002)
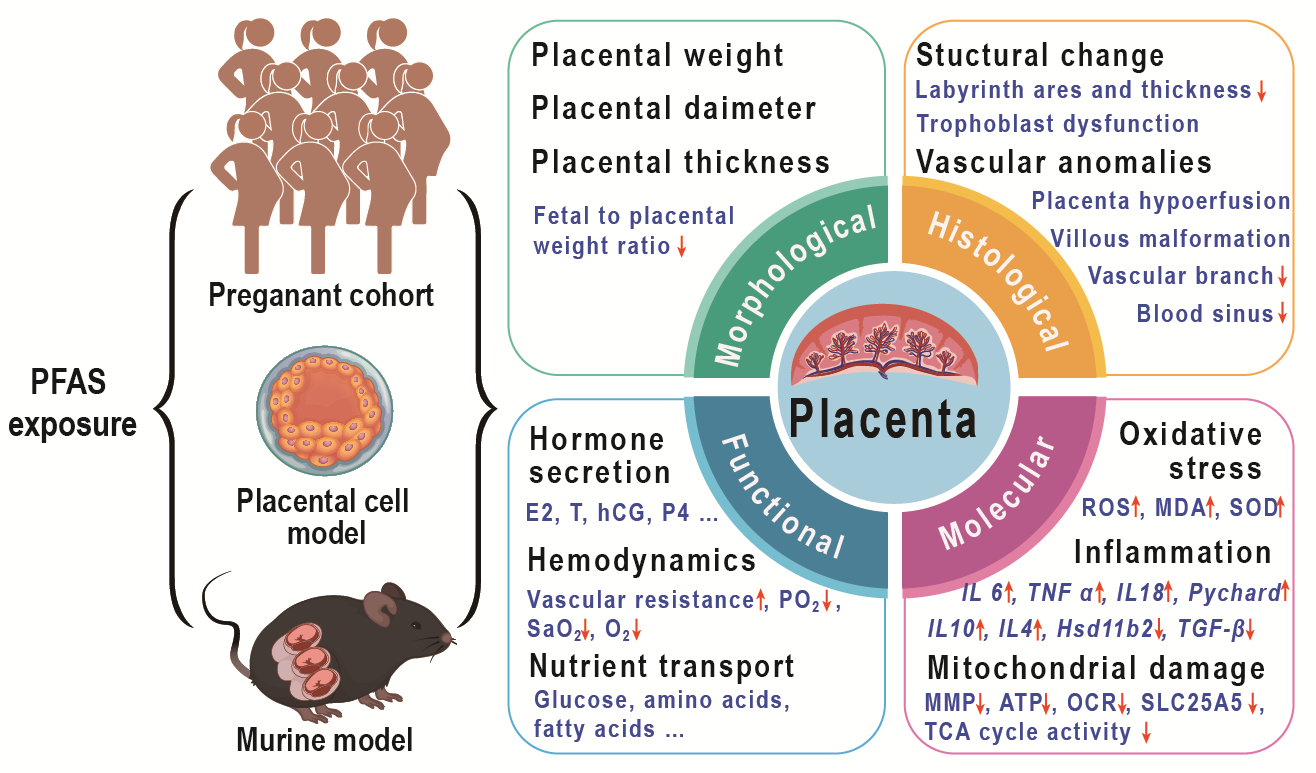
Per- and polyfluoroalkyl substances (PFAS) are a category of persistent organic pollutants (POPs) that are ubiquitously found across various environmental media, due to their extensive application in industrial processes and consumer products. These substances can infiltrate the human body through diet, drinking water, inhalation and skin contact, thereby posing potential risks to human health. The placenta, a critical organ at the maternal-fetal interface, is integral to material exchange and endocrine regulation, functioning as a natural barrier to shield the fetus from harmful external agents. Nonetheless, PFAS can cross the placental barrier, accumulate in placental tissues, and subsequently disrupt normal placental physiological functions, which poses significant threats to fetal growth and development. Based on evidence from epidemiological studies, placental cell models, and animal exposure models, this review summarizes the global exposure levels of PFAS in the placenta, examines the effects of PFAS exposure on placental morphology, structure, and function, and explores the underlying molecular mechanisms. By providing a comprehensive overview of current research, this review also offers insights into future research directions.
Contents
1 Introduction
2 Exposure of placentas to PFAS
2.1 Exposure concentration and distribution of human placentas to PFAS
2.2 Factors influence PFAS retention and transport in the Placenta
3 The effect of PFAS on placental structure and function
3.1 Effect on placental morphological
3.2 Effect on placental histological structure
3.3 Effect on placental vascular
3.4 Effect on nutrient metabolism and transport
3.5 Effect on placental endocrine
3.6 Molecular mechanisms of placental dysfunction
4 Conclusion and outlook
Key words: PFAS; placental structure; vessels; endocrine; nutrient transport; molecular mechanisms
Feng Quan , Chuanzi Gao , Wenhui Qiu , Yi Zheng . The Impact of Prenatal Perfluorinated and Polyfluoroalkyl Substances Exposure on Placental Structure and Function[J]. Progress in Chemistry, 2025 , 37(11) : 1719 -1730 . DOI: 10.7536/PC20250610
表1 各国的人群胎盘中的PFAS暴露浓度 (ng/g)Table 1 The concentration of PFAS (ng/g) in human placentas across different countries |
| Country | Region | Year | Sample size | Statistic | PFOA | PFOS | PFNA | PFHxS | PFDA | Ref |
|---|---|---|---|---|---|---|---|---|---|---|
| China | Tianjing | 2010 | 29 | Median | 1.41 | 7.32 | 0.96 | 0.36 | 0.67 | 15 |
| China | Fuxin | 2020 | 50 | Median | 21.00 | 2.50 | 8.40 | 0.06 | 1.80 | 33 |
| China | Guangzhou | 2021 | 302 | Median | 4.57 | 1.81 | 0.14 | <MDL | 0.17 | 36 |
| China | Wuhan | 2015-2016 | 32 | Median | 0.46 | 2.42 | / | 0.21 | / | 28 |
| China | Henan | 2016 | 54 | Median | 0.18 | 0.35 | 0.06 | 0.08 | <MDL | 32 |
| China | Henan | 2019-2020 | 48 | Median | 0.57 | 0.77 | 0.09 | <MDL | 0.07 | 40 |
| China | Hebei | 2013-2014 | 180 | Median | 1.33 | 1.21 | 0.23 | 0.05 | 0.21 | 39 |
| Unite States | North Carolina | 2010-2011 | 120 | Median | 0.27 | 0.95 | 0.11 | <MDL | 0.06 | 34 |
| Unite States | North Carolina | 2015-2018 | 122 | Median | 0.11 | 0.88 | 0.08 | 0.06 | 0.05 | 31 |
| Unite States | Arkansas | 2010-2014 | 151 | Median | 0.07 | 0.43 | 0.05 | 0.05 | 0.04 | 41 |
| Denmark | Denmark | / | 34 | Mean | 0.23 | 1.30 | 0.14 | <MDL | 0.10 | 29 |
| Sweden | Sweden | / | 38 | Mean | 0.41 | 1.40 | 0.18 | <MDL | 0.21 | 30 |
| Austria | Austria | 2017-2019 | 135 | Median | 0.22 | 0.34 | 0.06 | 0.03 | 0.03 | 37 |
| Spain | Granada | / | 20 | Median | 0.14 | 0.6 | 0.06 | 0.07 | 0.03 | 44 |
| Israel | Be'er Sheva | 2018-2021 | 50 | Median | 0.08 | 0.26 | 0.04 | 0.06 | 0.02 | 45 |
| Italy | Reggio Emilia | 2021-2022 | 20 | Median | 0.08 | 0.2 | / | 0.03 | / | 38 |
MDL is defined as method detection limit |
表2 关于PFAS与胎盘重量关联的流行病学研究Table 2 Epidemiologic studies on the associations between PFAS and placental weight |
| Author, Year | Country | Sample size | Results of placenta weight | Ref |
|---|---|---|---|---|
| Fei et al., 2008 | Denmark 1996-2002 | 1400 | Maternal PFOA and PFOS levels did not have any statistically significant effect on placental weight. | 51 |
| Gan et al., 2024 | China 2013-2014 | 712 | Positive associations found between br-PFHxS, 6∶2 Cl-PFESA, and placental weight. Mixed PFAS exposure (especially in 2nd/3rd trimesters) was positively linked to placental weight and scaling exponent (indicating reduced placental efficiency). | 52 |
| Chowdhury et al., 2024 | USA 2015-2019 | 158 | PFNA was inversely associated with placental weight (β=-0.17, 95% CI: -0.33, -0.019). PFDA showed a positive association with FPR. PFOA was negatively linked to placental arterial vascularization (mean distance from arterial end point to perimeter), but no direct effect on birth weight was observed. | 53 |
| Khan et al., 2025 | France 2014-2017 | 340 | Moderate-to-higher PFAS exposure cluster was associated with lower placental weight (-30 g, 95% CI: -56, -4.3), but no individual PFAS was directly associated with placental weight or FPR. | 54 |
表3 关于PFAS与胎盘重量关联的啮齿类动物模型研究Table 3 Rodent models on the associations between PFAS and placental weight |
| Author, Year | Animal species | Exposure doses | Duration of Exposure | Results of placenta weight | Ref |
|---|---|---|---|---|---|
| Suh et al., 2011 | CD-1 Mice | PFOA:0, 2, 10 and 25 mg/kg/day | GD 11 to 16 | Significantly reduced with increasing PFOA dose. | 55 |
| Lee et al., 2015 | CD-1 Mice | PFOS:0, 0.5, 2 and 8 mg/kg/day | GD 11 to 16 | Mean palcenntal weight was significantly reduced in all the treated groups and this reduction was seen in proportion to the PFOS dosage | 58 |
| Li et al., 2016 | Sprague-Dawley (SD) rats | PFOS: 0, 5 and 20 mg/kg/day | GD 12 to 18 | Significant reduction for the group treated with 20 mg PFOS | 57 |
| Blake et al., 2020 | CD-1 Mice | PFOA: 1 or 5 mg/kg/day; GenX: 2 or 10 mg/kg/day | GD 1.5 to 11.5 or 17.5 | GD11.5: No significant difference. GD17.5: placental weight and fetus-placenta weight ratioswas significantly higher in 5 mg/kg/d PFOA or 10 mg/kg/d GenX treated groups. | 56 |
| Zhang et al., 2023 | ICR mice | PFHxS: 0, 0.03, and 0.3 µg/kg/day | GD 7 to 17 | Placental weight significanty lower in high dose group | 59 |
| Adams et al., 2024 | CD-1 | PFOA: 0, 5, 100 ng/L; FTEOs: 0, 5, 100 ng/L (drinking water) | GD 0.5 to 17.5 | With a notable increase in placental weights in the 100 ng/L FTEOs group (13% for females, 15% for males). FTEOs exposure causing a larger decrease in the fetus-placental weight ratio compared to PFOA exposure. | 18 |
| Zhu et al., 2024 | CD-1 | PFDMO2HpA :0.01, 0.04, 0.16, 0.63, 2.5 mg/kg/day; PFDMO2OA 0.04, 0.16, 0.63, 2.5, 10 mg/kg/day | GD 2 to 12 or 18 | Gestational exposure to PFDMO2HpA/PFDMO2OA induced placenta enlargement, decreased fetus weight and placental efficiency | 60 |
图2 关于PFAS暴露对类固醇激素及其合成酶影响的主要研究结果总结,带箭头的蓝色方框表示相关激素水平上升或上调合成酶,带短横线的红色方框表示下降或下调,黄色连接线表示调控同一基因,黑色的文字代表已有研究验证的潜在机制,灰色字体代表尚未研究的潜在机制Fig.2 Summary of major findings on the effects of PFAS exposure on steroid hormones and steroidogenic enzymes. Blue boxes with arrows indicate increased levels of the corresponding hormone or enzyme, red boxes with lines indicate decreased levels, yellow connecting lines indicate the regulation of the same gene. Black text represents mechanisms that have been validated in previous studies, while gray text indicates potential mechanisms that have not yet been investigated |
| [1] |
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
Toxicological profile for perfluoroalkyls: United States. Agency for Toxic Substances and Disease Registry. 2021. [2025-06-01]http://dx.doi.org/10.15620/cdc:59198.
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
|
| [19] |
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
(鲍佳, 渠文娥, 邵立新, 刘洋, 王新, 吴雨晴. 中国环境科学, 2023, 43(7): 3730).
|
| [36] |
|
| [37] |
|
| [38] |
|
| [39] |
|
| [40] |
|
| [41] |
|
| [42] |
|
| [43] |
|
| [44] |
|
| [45] |
|
| [46] |
|
| [47] |
|
| [48] |
|
| [49] |
|
| [50] |
|
| [51] |
|
| [52] |
|
| [53] |
|
| [54] |
|
| [55] |
|
| [56] |
|
| [57] |
|
| [58] |
|
| [59] |
|
| [60] |
|
| [61] |
|
| [62] |
|
| [63] |
|
| [64] |
|
| [65] |
|
| [66] |
|
| [67] |
|
| [68] |
|
| [69] |
|
| [70] |
|
| [71] |
|
| [72] |
|
| [73] |
|
| [74] |
|
| [75] |
|
| [76] |
|
| [77] |
|
| [78] |
|
| [79] |
|
| [80] |
|
| [81] |
|
| [82] |
|
| [83] |
|
| [84] |
(朱天如, 曹静, 洪加威, 李静. 中华劳动卫生职业病杂志, 2020, 38(07):481).
|
| [85] |
|
| [86] |
|
| [87] |
|
| [88] |
|
| [89] |
|
| [90] |
|
/
| 〈 |
|
〉 |