

Application of UiO-66 Series MOFs in Proton Exchange Membranes
Received date: 2025-05-30
Revised date: 2025-07-16
Online published: 2025-12-10
Supported by
National Natural Science Foundation of China(22105225)
China State Key Laboratory of Heavy Oil Processing Research Fund(CNIF20250204)
Joint Research Institute for Carbon Neutrality(SKLHOP2024115806)
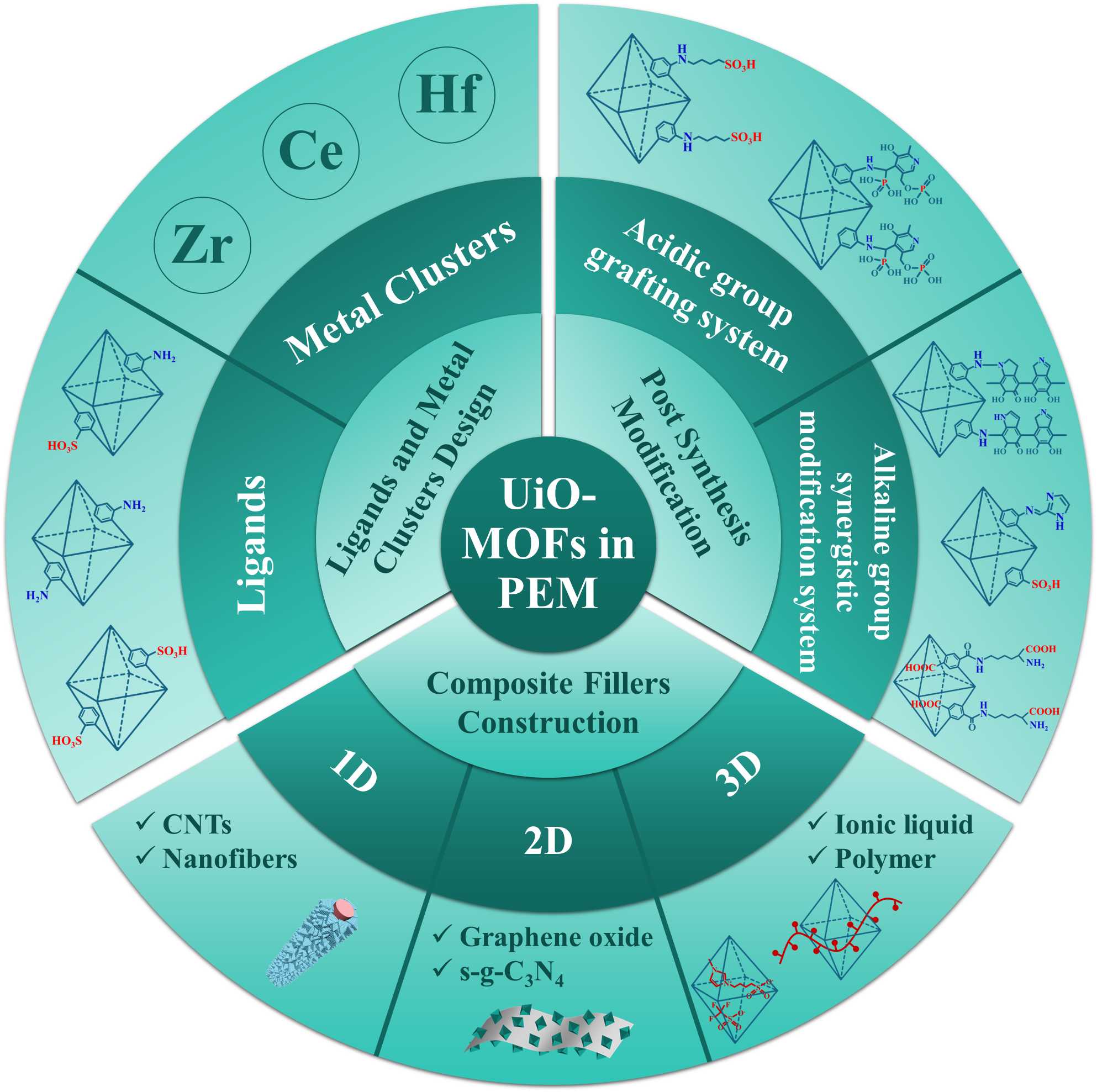
Metal-organic frameworks (MOFs) are emerging proton-conducting materials widely used in the modification of proton exchange membranes (PEM). Among them, the UiO-66 series MOFs (UiO-MOFs) exhibit high thermal and chemical stability, and are easy to synthesize and modify, making them ideal for PEM modification. This paper primarily reviews related research on UiO-MOFs used for PEM modification over the past five years from the perspective of filler design and preparation. Section II introduces the materials and proton conduction mechanisms of UiO-MOFs. Section III summarizes the design of ligands and metal clusters in UiO-MOFs, such as acid/base group modifications and metal cluster replacements. Section IV consolidates the methods for post-synthetic modifications of UiO-MOFs, such as grafting acid/base groups using active functional groups from external crystal structures. Section V presents various composite schemes involving UiO-MOFs and other materials to construct composite fillers with different dimensionalities. Finally, the summary highlights unresolved issues regarding the use of UiO-MOFs in PEMs and proposes future research directions.
1 Introduction
2 UiO-MOFs and proton conduction mechanism
2.1 UiO-MOFs material properties
2.2 Proton conduction mechanism
3 Design strategy of UiO-MOFs ligands and metal clusters
3.1 Ligand functionalization regulation strategy
3.2 Metal clusters regulation strategy
4 Post-synthetic modification of UiO-MOFs
4.1 Acid group grafting system
4.2 Alkaline group synergistic modification system
5 Construction of UiO-MOFs composite fillers
5.1 Construction strategy of 1D ordered composite fillers
5.2 Construction strategy of 2D composite fillers
5.3 Multi-dimensional control strategy for 3D composite fillers
6 Conclusion and future work
Mengxin Wang , Xiaocan Zhang , Qiong Zhou . Application of UiO-66 Series MOFs in Proton Exchange Membranes[J]. Progress in Chemistry, 2025 , 37(12) : 1731 -1757 . DOI: 10.7536/PC20250522
图2 (a) UiO-66结构以面心立方排列为特征,包括金属节点(浅绿色)和配体(灰色)。节点由与12个对苯二甲酸连接体连接的金属原子形成,形成两种笼状结构(12和7.5 Å)[32];(b) UiO-66简易图;(c) 不同晶体尺寸UiO-66的SEM图像[33];(d) MOFs功能化的不同策略示意图[37]Fig.2 (a) UiO-66 structure, characterized by a face-center-cubic arrangement, comprises metal nodes (aqua) and ligands (gray). The nodes are formed by metal atoms connected with 12 terepthalic acid linkers, resulting in two types of cage structures (12 and 7.5 Å)[32]; (b) schematic diagram of UiO-66; (c) SEM images of UiO-66 with different crystal sizes[33]; (d) schematic diagram of different strategies for the functionalization of MOFs[37]. Ref. [32] Copyright 2020 American Chemical Society. Ref. [33], Copyright 2017, American Chemical Society. Ref. [37], Copyright 2019, American Chemical Society |
图5 (a) 酸性基团改性UiO-MOFs简易图[54-57];(b) 嵌入Nafion基质中的S-U66和杂化膜中三种类型的质子传输通道的示意图[56];(c) 微嵌段聚合物链结构和胶束示意图及USO-15 wt%@SPP-3膜截面SEM图[59]Fig.5 (a) Schematic diagram of acidic group modified UiO-MOFs[54-57]; (b) schematic illustration of S-U66 embedded in Nafion matrix and three types of proton transport channels in hybrid membranes[56]; (c) schematic diagram of microblock polymer chain structures and micelles, along with SEM images of USO-15 wt%@SPP-3 membrane cross-sections[59]. Ref. [56], Copyright 2021, Elsevier. Ref. [59], Copyright 2025, Elsevier |
图6 (a) UiO-66-NH2简易图;(b) SUS三层膜结构示意图[64];(c) UiO-66-NH2-SPES复合膜的质子传导机理[66]Fig. 6 (a) Schematic diagram of UiO-66-NH2; (b) schematic of the SUS three-layer membrane structure[64]; (c) proton transfer mechanism of the UiO-66-NH2-SPES composite membrane[66]. Ref. [64], Copyright 2024, Elsevier. Ref. [66], Copyright 2023, American Chemical Society |
图7 (a) 复合膜内两类MOFs与聚合物三者之间的相互作用及膜的AFM相图[68];(b) NUS简易图和SPEEK/NUS-X复合膜中四类质子传输机理及膜截面SEM图[70]Fig.7 (a) The interactions among two types of MOFs and polymers within composite membranes and AFM phase diagram of membranes[68]; (b) schematic diagram of NUS as well as four types of proton transport mechanisms in SPEEK/NUS-X composite membranes and cross-sectional SEM images of membranes[70]. Ref. [68], Copyright 2017, American Chemical Society. Ref. [70], Copyright 2024, Elsevier |
图8 (a) 金属中心的元素选择;(b) 不同金属中心UiO-66的水吸附等温线和瞬态图及金属附近水分子的数量和瞬态图[76];(c) 金属中心的氟化改性与制备UN-X%或F-UN-X%@Nafion的流程图[78]Fig.8 (a) Element selection of metal clusters; (b) water adsorption isotherm and snapshots, as well as number and snapshots of water molecules near metal of UiO-66[76]; (c) fluorination modification of metal clusters and flow diagram for preparing UN-X% or F-UN-X%@Nafion[78]. Ref. [76], Copyright 2023, Elsevier. Ref. [78], Copyright 2021, Elsevier |
图10 (a) OPBI、PSM 2和纳米复合膜的XRD图(a1)及纯OPBI、PSM 1-10%和PSM 2-10%复合膜的SEM横截面图像(a2~a4),PSM 1-10% (a5)和PSM 2-10% (a6, a7)的TEM图像[79];(b) PUNSNPs/SPEEK=20%的截面SEM图[81];(c) SPEEK/UNC3S-3的STEM图像和SPEEK/NCC3S-3的高倍STEM-HAADF (c1)与元素分析(c2~c4)[82];(d) 染色PBIU-0.40 (d1)和UIO-66OSO3纳米颗粒之间密集离子簇(d2)的TEM图像[85]Fig. 10 (a) XRD patterns of OPBI, PSM 2 and nanocomposite membranes (a1), SEM cross-sectional images of pristine OPBI, PSM 1-10% and PSM 2-10% composite membranes (a2~a4), TEM images of PSM 1-10% (a5) and PSM 2-10% (a6, a7)[79]; (b) SEM cross-section images of PUNSNPs/SPEEK=20%[81]; (c) STEM image of SPEEK/UNC3S-3, high magnification STEM-HAADF (c1) and elemental analysis of SPEEK/UNC3S-3 (c2~c4)[82]; (d) TEM image of stained PBIU-0.40 (d1) and densely ionic clusters between UIO-66OSO3 nanoparticles (d2)[85]. Ref. [79], Copyright 2020, American Chemical Society. Ref. [81], Copyright 2023, Elsevier. Refs. [82,85], Copyright 2022, Elsevier |
图11 各类碱性基团协同修饰的UiO-MOFs结构简图:(a) 咪唑基团修饰的UiO-66[89-91];(b) 氨基酸官能化的Glu-UiO-66-(COOH)2和Lys-UiO-66-(COOH)2[93];(c) UN@PDA[94];(d) 聚合物刷修饰的MOFs[95]Fig.11 Schematic diagram of UiO-MOFs co-modified by alkaline synergistic modification. (a) UiO-66 modified with imidazole groups[89-91]; (b) amino acid-functionalized Glu-UiO-66-(COOH)2 and Lys-UiO-66-(COOH)2[93]; (c) UN@PDA[94]; (d) polymer brush modified MOFs[95] |
图12 (a) CC/S-UIO-66@CNT膜的质子传导机理和膜的AFM表面形貌,(a1, a2) CNF/CNC, (a3, a4) CC/S-UIO-66@CNT-5[97];(b) UiO-66-NH2@NFs复合膜的设计结构和可能存在的质子传输途径及其表面与截面SEM图像[99];(c) 同轴静电纺丝和制备工艺示意图与膜的AFM相图[101];(d) 膜制备过程和质子转移机制及(d1, d2) 1.3% NU6@PPNF-SPEEK的STEM-HAADF图和元素分析,(d3, d4)SPEEK和5% NU6-SPEEK的原位FTIR(从180 ℃冷却到30 ℃)[104]Fig.12 (a) Proton conduction mechanism of CC/S-UIO-66@CNT membranes and AFM surface topographies of (a1, a2) CNF/CNC, (a3, a4) CC/S-UIO-66@CNT-5[97]; (b) designed structure of UiO-66-NH2@NFs and possible routes for proton transfer in the hybrid membranes, as well as surface and cross-sectional SEM images[99]; (c) schematic diagram of the coaxial electrospinning and preparation process and AFM phase images of membranes[101]; (d) membrane preparation process and proton transfer mechanism, as well as (d1, d2) STEM-HAADF image and elemental analysis of 1.3% NU6@PPNF-SPEEK, (d3, d4) in-situ FTIR of SPEEK and 45% NU6-SPEEK (cooling process from 180 ℃ to 30 ℃)[104]. Ref. [97], Copyright 2025, American Chemical Society. Ref. [99], Copyright 2019, Elsevier. Ref. [101], Copyright 2024, Elsevier. Ref. [104], Copyright 2021, Elsevier |
图13 (a) SPEEK/S-UiO-66@GO复合膜增强的传输特性[111];(b) UiO-66/Pd-GO和复合膜的结构示意图与填料的电子能带结构[113];(c) CS/U-S@GO膜质子传导机理[115]Fig.13 (a) Enhanced transport properties of SPEEK/S-UiO-66@GO composite membranes[111]; (b) schematic diagram of UiO-66/Pd-GO and composite membrane structure, as well as electronic band structure of fillers[113]; (c) proton transfer mechanism of CS/U-S@GO membrane[115]. Ref. [111], Copyright 2017, American Chemical Society. Ref. [113], Copyright 2021, American Chemical Society. Ref. [115], Copyright 2020, Wiley |
图14 三维复合填料示意图:(a) UiO-66-AS@ILs[119];(b) S-UiO[121];(c) UiO-67 MOF/TAPB-DMTP-COFs(Schiff碱缩合反应)核壳MOFs合成方法[123]Fig.14 Schematic diagram of 3D composite fillers: (a) UiO-66-AS@ILs[119] and (b) S-UiO[121]; (c) UiO-67 MOF/TAPB-DMTP-COFs (Schiff base condensation reaction) core shell MOFs synthetic root[123]. Copyright 2024, Elsevier |
表1 不同改性手段UiO-MOFs用于PEM的优缺点总结Table 1 Summary of advantages and disadvantages of using UiO-MOFs with different modification methods for PEM |
| Methods | Advantages | Disadvantages |
|---|---|---|
| Pre-synthetic modification (ligand functionalization or metal clusters modification) | For MOFs: √ One-step synthesis method. √ Modulation of metal ions or organic ligands allows for flexible and convenient regulation of the textural properties. √ Adjustable size and strong stability. √ Evenly distributed functional groups inside the crystal facilitate orderly and unobstructed internal channels. √ High functional group density modified upon pre-synthesis. √ Metal clusters: Zr: Low price. Ce: Functionalized in eliminating free radicals. Hf: High Hf-O bond energy. | For MOFs: × Low conductivity. × Difficult to adjust or replace functional groups modified before synthesis. × Metal clusters: Zr: slightly lowered Zr-O bond energy. Ce: slightly less stable and higher cost. Hf: highest cost. |
| For PEM: √ Introducing proton sources, proton hopping sites, or acid-base pairs. √ Increase water absorption. √ The hydrogen bond network inhibits swelling. | For PEM: × Relatively low conductivity. × An increase in water absorption may accelerate polymer degradation. | |
| Post-synthetic modification (grafting acidic or alkaline functional groups) | For MOFs: √ External crystal modification to preserve the complete skeleton. √ Flexible design. √ High conductivity. | For MOFs: × Complex synthesis and lower yields. × Synthesis conditions may damage the crystal structure. × Uneven distribution of functional groups or local overload. × Low functional group density modified upon post-synthesis. |
| For PEM: √ Introducing proton sources, proton hopping sites, or acid-base pairs. √ Improvement of compatibility with substrates. | For PEM: × Complex process. × High cost. | |
| Construction of UiO-MOFs composite filler | For MOFs: √ Orderly distributed. √ Well dispersed. √ Intra-pore modified. | For MOFs: × Complex construction of composite fillers. × Uneven distribution of modifications within the pores. × Limited types of substances loaded into MOFs due to the pore size. |
| For PEM: √ Enhance mechanical strength. √ Reduce MOFs aggregation. √ Arrange directional MOFs to construct long-range ordered proton transport channels. √ 2D topography decreases fuel penetration. | For PEM: × Leakage of small-molecule acids causes device corrosion. |
表2 不同UiO-MOFs质子交换膜性能总结Table 2 Summary of the performance of different UiO-MOFs proton exchange membranes |
| MOFs | Composition [Type a, thickness (μm)] and polymer [DS b] | Proton conductivity (×10-3 S/cm), swelling ratio and water uptake/ADL c | Battery performanced | Tensile strength (MPa) | Long term stability e | Ref | ||
|---|---|---|---|---|---|---|---|---|
| 1. UiO-MOFs without functional group modification | ||||||||
| UiO-66 | N_U200-2 [LT, 100] Nafion [EW=1100] | 165 | - | - | - | 12.9±0.4 | - | 45 |
| 95%RH) | ||||||||
| S/UiO-66 [VRFB, 57] SPEEK | ~28 | - | - | CE=97.5% EE=90.9% | - | 100 cycles 0.18% (CD) | 46 | |
| 100%RH) | ||||||||
| 50-sPBI-UiO-5 [VRFB, 15] sPBI [43.3%] | 17.8 | 6.5% | 10.6% | CE=97.09% EE=82.84% | 70.5±2.9 | 50 cycles 1.2% (CD) | 47 | |
| 100%RH) | ||||||||
| SP20/12.5MOF [LT, -] SPEEK [60%], PBI | 192 | - | 45% | - | - | - | 48 | |
| 100%RH) | ||||||||
| 40%UIO-66@OPBI [HT, 69.70±0.74] OPBI | 92 | 63.13% (PA) | 3.09 | 583 (160 ℃, 0%RH) | 27.02±1.51 | 500 h 0.15 (OCV) | 49 | |
| (160 ℃, 0%RH) | ||||||||
| CBOPBI@MOF40% [HT, 45] CBOPBI | 100 | 50% (PA) | 126% (PA) | 607 (160 ℃, 0%RH) | 8.1±0.4 | 187 h 0.036 (OCV) | 50 | |
| (160 ℃, 0%RH) | ||||||||
| UIO-66@OPBI [HT, 63±2] OPBI | 88 | 139.41% (PA) | 211.20% (PA) | 546.62 (160 ℃, 0%RH) | >15 | - | 52 | |
| (160 ℃, 0%RH) | ||||||||
| PBI-UiO66 (10.0 wt %) [HT, 70-100] PBI | 316 | - | 11.0 | - | 5.4±0.4 | - | 51 | |
| (160 ℃, 0%RH) | ||||||||
| 2. UiO-MOFs modified with acidic groups only | ||||||||
| UiO-66-(COOH)2 | UiO-66-(COOH)2@PP-30 [LT, -] PVDF, PVP | 5.8 | 1.8% | 58.9% | - | - | 7 d >90% (σ) | 54 |
| 98%RH) | ||||||||
| UiO-66-SO3H | sPSF/sUiO-66 3wt% [LT, -] sPSF | 180 | 28% | 73% | - | 163 | - | 57 |
| 100%RH) | ||||||||
| Nf/S-U66-3 [VRFB, 52] Nafion | ~44 | 7.98% | 20.60% | CE=97.0% EE=76.1% | 4.02 | 230 cycles 0.13% (CD) | 56 | |
| 100%RH) | ||||||||
| UiO-66-(SO3H)2 | USO-15wt%@SPP-3 [LT, VRFB, 28] SPP-3 | 165.0 | 23.3% | 25.7% | 387.7 (80 ℃, 100%RH) | 62.6 | - | 59 |
| 100%RH) | ||||||||
| UiO-66 modified with —PO3H2 | 1:2G-UIO-66@OPBI [HT, 52.36±0.37] OPBI | 124 | 99.46% (PA) | 72.58% (PA) | 725 (160 ℃, 0%RH) | 20.88±0.99 | 200 h 0.042 (OCV) | 55 |
| (160 ℃, 0%RH) | ||||||||
| 3. UiO-MOFs modified with alkaline groups only | ||||||||
| UiO-66-NH2 | SUS5 [LT-HT, 30] Nafion | 428 | 1.58% | 30.7% | 940 (115 ℃, 100%RH) | ~16 | 132 h 91.1% (σ) | 64 |
| 100%RH) | ||||||||
| Am-S-U-6% [LT, -] Am-SPAEKS | 168 (80 ℃) | 9.49% | 56.72% | 290.97 (80 ℃, 100%RH) | 55.26±0.50 | 240 h 95.6% (σ) | 67 | |
| 98%RH) | ||||||||
| 3% CM [LT, -] SPES [~17%] | 214.9 | ~17% | ~18% | 180 (25 ℃, 0%RH) | 22.41 | - | 66 | |
| 98%RH) | ||||||||
| SPEN/UiO-66-NH2-5 [LT, -] SPEN | 135.1 | ~11% | ~60% | - | 60.8 | - | 62 | |
| 100%RH) | ||||||||
| CBOPBI-P40 [HT, 65] CBOPBI | 84 | 55% (PA) | 296% (PA) | 750 (180 ℃, 0%RH) | ~8.5 (PA) | 387 h 0.010 (OCV) | 53 | |
| (160 ℃, 0%RH) | ||||||||
| CUN15@OPBI [HT, 30-40] OPBI (carbonized MOFs) | 79 | 117.93% (PA) | 229.45% (PA) | 952 (160 ℃, 0%RH) | ~12 (PA) | 10 h >99% (σ) | 65 | |
| (180 ℃, 0%RH) | ||||||||
| MU-NH2/Nf-3 [VRFB, 40±1] Nafion | 122.18 | ~3% | ~20% | CE=97.9% EE=83.8% | 27.3 | 200 cycles 0.19% (CD) | 60 | |
| 100%RH) | ||||||||
| 4. UiO-MOFs modified by both acidic and basic groups | ||||||||
| UiO-66-NH2 and UiO-66-SO3H | UiO-66-NH2+UiO- 66-SO3H/Nafion-0.6 [LT, 72]; Nafion | 256 | ~14% | ~29% | - | - | 50 h ~98% (σ) | 68 |
| 95%RH) | ||||||||
| CS/A-6 + B-15 [HT, 200-400] Chitosan | 3.78 | - | - | 10.6 (120 ℃, 0%RH) | - | - | 69 | |
| (120 ℃, 0%RH) | ||||||||
| NH2-UiO-66-SO3H | SPEEK/NUS-1.5 [LT, 100] SPEEK [54.1%] | 177.76 | ~30% | 41.86% | 423.2 (75 ℃, 100%RH) | 50.91 | 300 h 89.3% (σ) | 70 |
| 100%RH) | ||||||||
| 5. UiO-MOFs with different metal clusters | ||||||||
| SA-Hf-UiO-66-(OH)2 | CS/SA-1-6 [LT, -] Chitosan | 21.9 | ~60% (25 ℃) | ~60% (25 ℃) | - | ~8 | - | 75 |
| 98%RH) | ||||||||
| Ce-UiO-66 | Ce-3 [water electrolysis, -] Nafion [EW=1030-1120] | 124.45 | ~3% | ~11.5% | Electrolysis efficiency=65.4% | 15.24 | - | 76 |
| 90%RH) | ||||||||
| d-UiO-66(Ce) | PFSA/d-UiO-66 [12] (spraying) Nafion | - | - | - | 1960 | - | 100 h | 77 |
| - | ||||||||
| F-UN | F-UN-5.0%@Nafion [LT, 30-50] Nafion | ~240 | ~26% | ~39% | 26.8 | 35.8 | 387 h 0.78 (OCV) | 78 |
| 100%RH) | ||||||||
| 6. Acid group grafting system | ||||||||
| PUIN | FSPUIN-5 [LT, -] F-SPAEKS | 174.22 | 13.76% | 18.25% | 957 (80 ℃, 100%RH) | 30.70±1.47 | 25 h 4 (OCV) | 87 |
| 100%RH) | ||||||||
| SPUIN | FSSPU-7 [LT, -] F-SPAEKS | 247.38 | 16.60% | 33.96% | 1140 (80 ℃, 100%RH) | 32.37±3.01 | 64 h 1.73 (OCV) | 88 |
| 100%RH) | ||||||||
| PVP-UiO-66-NH-SO3H | PA/PUNSNPs/SPEEK=20 % [LT, -] SPEEK [68%] | 350 | ~10% | ~50% | - | - | 40 d >83% (σ) | 81 |
| 100%RH) | ||||||||
| UNCS | SPEEK/UNCS-3 [LT, -] SPEEK [42%] | 186.4 | ~26% | ~45% | - | 61.9 | - | 82 |
| 100%RH) | ||||||||
| UiO-66-NH-SO3H | UiO-66-NH-SO3H/SPEEK=30% [LT, -] SPEEK [68%] | 273 | ~19% | ~48% | - | ~90 | - | 83 |
| 100%RH) | ||||||||
| PSM 2 | PSM 2-10% [HT, -] OPBI | 308 | 4.57% (PA) | 31.35 | - | 1.18 (PA) | 24 h 89.1% (σ) | 79 |
| (160 ℃, 0%RH) | ||||||||
| UIO-66OSO | PBIU-0.40 [VRFB, 30] PBI | 434.8 | <6.7% | ~30% | CE=99.3% EE=86.1% | >70.2 | 100 cycles 0.15% (CD) | 85 |
| 100%RH) | ||||||||
| PA-UiO-66-NH2 | S/PA-UiO-66-NH2-2 [VRFB, 62] SPEEK | 35.3 | 9.3% | 32.4% | CE=99.1% EE=83.8% | 32.7 | 100 cycles 0.60% (CD) | 86 |
| 100%RH) | ||||||||
| 7. Alkaline group synergistic modification system | ||||||||
| IUSN | C-SPAEKS/IUSN-3% [LT,30-40] C-SPAEKS | 234 | 29.37% | 44.24% | 243.77 (80 ℃, 100%RH) | 48.67 | 240 h 97% (σ) | 90 |
| 100%RH) | ||||||||
| IM-UIO-66-AS | IM-UIO-66-AS/SPEEK [VRFB, 30] SPEEK | 12.2 | ~10% | ~29% | CE=99.6% EE=79.6% | ~47 | 100 cycles 0.26% (CD) | 89 |
| 100%RH) | ||||||||
| UiO-66-IM | 10% UiO-66-IM/6FPBI [HT, 50±5] 6FPBI | 41.8 | - | 16.5 | 406.1 (160 ℃, 0%RH) | 93.3±10.1 | - | 91 |
| (170 ℃, 0%RH) | ||||||||
| UiO-66-NH2-Glu | UiO-66-NH2-Glu/SPSF [LT, 80] SPSF | 212 | ~19% | ~39% | 70.45 (60 ℃, 100%RH) | - | - | 92 |
| 100%RH) | ||||||||
| Lys-UiO-66-(COOH)2 | Lys-UiO-66-(COOH)2@CS-7 [LT, 400-500 (±60)]; Chitosan | 22 | - | - | - | ~18 | 240 h >98% (σ) | 93 |
| 100%RH) | ||||||||
| UN@PDA | SPEEK/UN@PDA-0.75 [VRFB, 80-85] SPEEK [71.0%] | 31.1 | ~10% | ~16% | CE=98.5% EE=81.6% | ~33 | 100 cycles 0.59% (CD) | 94 |
| 100%RH) | ||||||||
| Grafting of polymer brushes | OPBI@PGM-Z5% [HT, -] OPBI | 241 | 4.18% (PA) | 24.98 | - | ~1 (PA) | 24 h ~88% (σ) | 95 |
| (160 ℃, 0%RH) | ||||||||
| 8. Construction strategy of 1D ordered composite fillers | ||||||||
| UiO-66-NH2/CNT | SPEN@UiO-66-NH2/CNT-0.7 [LT, 60-80], SPEN | 173.7 | ~14.5% | ~47% | - | 32.48 | - | 96 |
| 100%RH) | ||||||||
| S-UIO-66@CNT | CC/S-UIO-66@CNT-5 [LT, 52] Cellulose | 105 | 1.4% | 53% | 3.56 (80 ℃, 100%RH) | 93.60 | 72 h 0.26 (OCV) | 97 |
| 100%RH) | ||||||||
| SO3H-UiO-66@HNTs | CS/SO3H-UiO-66@HNTs-10 [LT, -] Cellulose | 46.2 | 60% | 83% | 84.5 (70 ℃, 100%RH) | 57.1 | 100 h 0.682 (OCV) | 98 |
| 100%RH) | ||||||||
| UiO-66-NH2@NFs | UiO-66-NH2@NFs-8/Nafion [LT, ~70] Nafion; SPES [64%, NFs] | 270 | 29.20% | 37.32% | 95.49 (60 ℃, 100%RH) | 32.35 | 54 h ~99% (σ) | 99 |
| 100%RH) | ||||||||
| Cell-UiO-66-NH2 | Cell-UiO-66-NH2-5/SPSF [LT, 80-100] SPSF [60%] | 196 | ~17.5% | ~38% | 78 (60 ℃, 100%RH) | - | 130 h 86.7% (σ) | 100 |
| 100%RH) | ||||||||
| PU@S-PAN/UiO-66 | Nafion/S@NF-50 [LT, -] Nafion | 212 | ~19% | ~68% | 182.6 (65 ℃, 100%RH) | - | - | 101 |
| 100%RH) | ||||||||
| NU6@PPNF | 1.3% NU6@PPNF-SPEEK [LT, 96] SPEEK | 132.8 | 14.0% | ~23% | 175.7 (60 ℃, 95%RH) | >25 | 144 h 92.9% (σ) | 104 |
| 100%RH) | ||||||||
| SU6@PPNF | SU6@PPNF-SPEEK [LT, -] SPEEK | 154.6 | ~31% | ~50% | 172.1 (60 ℃, 100%RH) | - | - | 105 |
| 100%RH) | ||||||||
| NU6@PI | 4NP-SPEEK-4NP [LT, 120] SPEEK [58.3%] | 178.1 | 15.8% | ~29% | 217.01 (60 ℃, 95%RH) | 65.8 | 32 d 89.8% (σ) | 106 |
| 100%RH) | ||||||||
| NU6@PI | NU6@PI/SPEEK+HPW-60 [LT, 136.0±0.5]; SPEEK [55.6%] | 174.9 | 19.4% | 37.9% | 186.7 (60 ℃, 95%RH) | ~62 | 42 d 86.5% (σ) | 107 |
| 100%RH) | ||||||||
| UiO-66-NH2@SFM | UiO-66-NH2-8@SFM/Nafion [LT, -] Nafion | 286 | 12% | 52% | 88.3 (60 ℃, 100%RH) | 31.64 | - | 108 |
| 100%RH) | ||||||||
| UiO-66-NH2 +Caffeic acid | PPUC-Nafion-3 [LT, 24.3] Nafion | 212 | ~9% | ~40% | 812.64 (80 ℃, 100%RH) | ~14 | 100 h 1.11 (OCV) | 109 |
| 100%RH) | ||||||||
| PVDF/UiO NFMs | A-PVDF-NS@Nafion [LT, -] Nafion | 152.11 | ~8.5% | ~36% | - | 12.12 | - | 110 |
| 100%RH) | ||||||||
| 9. Construction strategy of 2D composite fillers | ||||||||
| S-UiO-66@GO | SPEEK/S-UiO-66@GO-10 [LT, 40±10] SPEEK [62%] | 268 | - | ~32% (30 ℃) | - | 53.5 | - | 111 |
| 95%RH) | ||||||||
| GO@UiO-66-NH2 | GO@UiO-66-NH2/Nafion-0.6 [LT, -] Nafion | 303 | ~10% | ~28% | - | - | 54 h ~99% (σ) | 112 |
| 95%RH) | ||||||||
| UiO-66/Pd-GO | SU3%/P10%-G2.5% [LT, -] SPEEK | 211 | - | 50.3% | - | - | - | 113 |
| 100%RH) | ||||||||
| UiO-66-NH2 and s-g-C3N4 | SPEEK/NF-1:1 [VRFB, 82] SPEEK [71%] | 27.9 | 11.4% | 15.1% | CE=98.8% EE=79.9% | 37.8 | 100 cycles 0.56% (CD) | 114 |
| 100%RH) | ||||||||
| UiO-66-SCNT@GO | CS/U-S@GO-7 [LT, 400-600] Chitosan | 64 | 1.01% | 79.85% | - | 50.73 | - | 115 |
| 100%RH) | ||||||||
| 10. Multi-dimensional control strategy for 3D composite fillers | ||||||||
| IL@UiO-66 | IL@UiO-66@PP [LT, 201.7] PVDF, PVP | 16.8 | - | 18.93% | - | 4.62 | 48 h ~99% (σ) | 116 |
| 98%RH) | ||||||||
| IL-modified UiO-66 | CBOPBI@MOF50%-IL30 [HT, 40-50] CBOPBI | 135 | 70% (PA) | 10.6 | 736 (160 ℃, 0%RH) | ~7.5 | 194 h 0.2 (OCV) | 117 |
| (160 ℃, 0%RH) | ||||||||
| Zr-MOF/IL | SMOF/TEA2.5 [LT, -] SPEEK [66%] | 140 | - | 94% | - | - | - | 118 |
| 60%RH) | ||||||||
| UiO-66-AS@ILs | C-S-U-AS@ILs-5% [LT, ~40] C-SPAEKS | 197 | 3.53 (80 ℃) | 19.3% (80 ℃) | 474.24 (80 ℃, 100 %RH) | 38.51±3.46 | - | 119 |
| 100%RH) | ||||||||
| S-UiO | SPEEK/S-UiO-15 [VRFB, 58] SPEEK | 67 | ~24% | ~39% | CE=99.5% EE=83.9% | 55.1 | 200 cycles 0.11% (CD) | 121 |
| 100%RH) | ||||||||
| UiO-67/TAPB-DMTP-COFs | PASCM-0.75 [HT, 80-90] SPEEK [70%] | 7.7 | 29% (PA) | 7.3 | - | 27.3 | - | 123 |
| (130 ℃, 0%RH) | ||||||||
| PAT@UiO-66-SO3H | CS/ PAT@UiO-66-SO3H-3 [LT, -] Chitosan | 38.8 | 53% (60 ℃) | 67% (60 ℃) | 37.9 (80 ℃, 100 %RH) | ~53 | - | 124 |
| 100%RH) | ||||||||
| UiO-66-NH2 and CDs | (SPEEK/40%CDs@MOF)3/PA [HT, ~65]; SPEEK [76.6%] | 50.2 | 37.8% (PA) | 4.9 | 369.9 (120 ℃, 0%RH) | ~8 | 200 h ~44% (σ) | 125 |
| (160 ℃, 0%RH) | ||||||||
a LT: low temperature PEM for fuel cells; HT: high temperature PEM for fuel cells; VRFB: vanadium redox flow battery.b DS: Sulfonation degree of polymer substrate materials.c High temperature PEM refers to swelling ratio and acid doping level (ADL) after doped phosphoric acid (PA), while others are in water.d Applied to fuel cells, the numerical value represents the maximum power density (mW/cm2). Applied to VRFB, the numerical values represent coulombic efficiency (CE) and energy efficiency (EE).e The retention rate of performance under the longest testing period. In parentheses, σ represents proton conductivity, CD represents average discharge capacity decay (per cycle) of VRFB and OCV represents the cell open-circuit voltages declay rate (mV/h) of the membrane electrode assemblies. |
表3 UiO-MOFs的质子电导率Table 3 Proton conductivity of UiO-MOFs |
| MOFs | Proton conductivity (S/cm) | Test conditions | Ref |
|---|---|---|---|
| 1. Pre-synthetic modification | |||
| UiO-66(Zr) | 7.54×10-6 | 30 ℃, 97%RH | 129 |
| UiO-66(Zr) (low-crystallinity MOG) | 1.23×10-2 | 80 ℃, 75%RH | 130 |
| UiO-66-NH2 | 1.40×10-5 | 30 ℃, 97%RH | 129 |
| UiO-66-SO3H | 0.34×10-2 | 30 ℃, 97%RH | 129 |
| UiO-66-(SO3H)2 | 8.4×10-2 | 80 ℃, 90%RH | 131 |
| UiO-66-(SO3H)4 | 3.7×10-1 | 90 ℃, 90%RH | 132 |
| UiO-66-COOH (powder) | 4.99×10-2 | 80 ℃, 100%RH | 133 |
| UiO-66-COOH (gel) | 8.53×10-2 | 80 ℃, 100%RH | 133 |
| UiO-66-(COOH)2 | 0.10×10-2 | 30 ℃, 97%RH | 129 |
| H2SO4@UiO-66-SO3--NH3+ | 5.40×10-1 | 90 ℃, 100%RH | 134 |
| H2N-UiO-66-SO3H | 2.8×10-2 | 80 ℃, 75%RH | 135 |
| H2N-UiO-66-COOH | 4.1×10-4 | 80 ℃, 75%RH | 135 |
| HOOC-UiO-66-SO3H | 5.3×10-4 | 80 ℃, 75%RH | 135 |
| 2. Metal cluster modification | |||
| UiO-66(Ce) | 1.63×10-5 | 100 ℃, 98%RH | 136 |
| UiO-66-(Ce)-COOH | 2.25×10-4 | 100 ℃, 98%RH | 136 |
| UiO-66-(Ce)-(COOH)2 | 1.10×10-3 | 100 ℃, 98%RH | 136 |
| UiO-66(Ce)-Br2 | 0.61×10-3 | 100 ℃, 98%RH | 137 |
| UiO-66(Hf)-(OH)2 | 4.33×10-3 | 100 ℃, 98%RH | 138 |
| UiO-66(Hf)-NH2 | 1.10×10-3 | 100 ℃, 98%RH | 138 |
| UiO-66(Hf)-NO2 | 0.89×10-3 | 100 ℃, 98%RH | 139 |
| UiO-66(Hf)-COOH | 2.83×10-3 | 100 ℃, 98%RH | 140 |
| UiO-66(Hf)-(COOH)2 | 4.35×10-3 | 100 ℃, 98%RH | 140 |
| 3. Post-synthetic modification | |||
| UiO-66-NH2-g-1,3-PS | 1.64×10-1 | 80 ℃, 95%RH | 141 |
| UiO-66-NH2-g-1,4-BS | 4.6×10-3 | 80 ℃, 95%RH | 141 |
| IM-UiO-66-AS (imidazole) | 1.54×10-1 | 80 ℃, 98%RH | 142 |
| DT-UiO-66 (triazole) | 4.47×10-3 | 100 ℃, 100%RH | 143 |
| SO3H-UiO-66-N4 (tetrazole) | 5.5×10-5 | 85 ℃, 95%RH | 144 |
| UiO-66-COOH-Asp (aspartic acid) | 1.19×10-2 | 70 ℃, 98%RH | 145 |
| 60-UiO-66-1.8 (3-mercaptopropionic acid) | 3×10-2 | 100 ℃, 98%RH | 146 |
| PGM-L3 (poly(vinyl phosphonic acid)) | 1.26×10-2 | 80 ℃, 98%RH | 147 |
| UiO-66-SB (flexible zwitterionic arm) | 1.0×10-3 | 85 ℃, 90%RH | 148 |
| LiCl@UiO-66-F2(SO3H)2 | 2.86 | 90 ℃, 90%RH | 149 |
| 4. Defect engineering strategy | |||
| UiO-66(Hf)-NH2-3.18 (p-Aminobenzoic acid) | 4.56×10-2 | 90 ℃, 98%RH | 150 |
| D-UiO-66-N=IM (acetic acid + imidazole) | 2.15×10-2 | 70 ℃, 100%RH | 151 |
| Zr4+ terephthalate UiO-66 | 6.93×10-3 | 65 ℃, 95%RH | 152 |
| [1] |
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
|
| [19] |
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
|
| [36] |
|
| [37] |
|
| [38] |
|
| [39] |
|
| [40] |
|
| [41] |
|
| [42] |
|
| [43] |
|
| [44] |
|
| [45] |
|
| [46] |
|
| [47] |
|
| [48] |
|
| [49] |
|
| [50] |
|
| [51] |
|
| [52] |
|
| [53] |
|
| [54] |
|
| [55] |
|
| [56] |
|
| [57] |
|
| [58] |
|
| [59] |
|
| [60] |
|
| [61] |
|
| [62] |
|
| [63] |
|
| [64] |
|
| [65] |
|
| [66] |
|
| [67] |
|
| [68] |
|
| [69] |
|
| [70] |
|
| [71] |
|
| [72] |
|
| [73] |
|
| [74] |
|
| [75] |
|
| [76] |
|
| [77] |
|
| [78] |
|
| [79] |
|
| [80] |
|
| [81] |
|
| [82] |
|
| [83] |
|
| [84] |
|
| [85] |
|
| [86] |
|
| [87] |
|
| [88] |
|
| [89] |
|
| [90] |
|
| [91] |
|
| [92] |
|
| [93] |
|
| [94] |
|
| [95] |
|
| [96] |
|
| [97] |
|
| [98] |
|
| [99] |
|
| [100] |
|
| [101] |
|
| [102] |
|
| [103] |
|
| [104] |
|
| [105] |
|
| [106] |
|
| [107] |
|
| [108] |
|
| [109] |
|
| [110] |
|
| [111] |
|
| [112] |
|
| [113] |
|
| [114] |
|
| [115] |
|
| [116] |
|
| [117] |
|
| [118] |
|
| [119] |
|
| [120] |
|
| [121] |
|
| [122] |
|
| [123] |
|
| [124] |
|
| [125] |
|
| [126] |
|
| [127] |
|
| [128] |
|
| [129] |
|
| [130] |
|
| [131] |
|
| [132] |
|
| [133] |
|
| [134] |
|
| [135] |
|
| [136] |
|
| [137] |
|
| [138] |
|
| [139] |
|
| [140] |
|
| [141] |
|
| [142] |
|
| [143] |
|
| [144] |
|
| [145] |
|
| [146] |
|
| [147] |
|
| [148] |
|
| [149] |
|
| [150] |
|
| [151] |
|
| [152] |
|
/
| 〈 |
|
〉 |