

Construction Methods and Application Progress of Liquid-Like Surfaces
Received date: 2025-06-13
Revised date: 2025-07-13
Online published: 2025-12-10
Supported by
National Natural Science Foundation of China(22378253)
Natural Science Basic Research Program of Shaanxi(2024JC-YBMS-122)
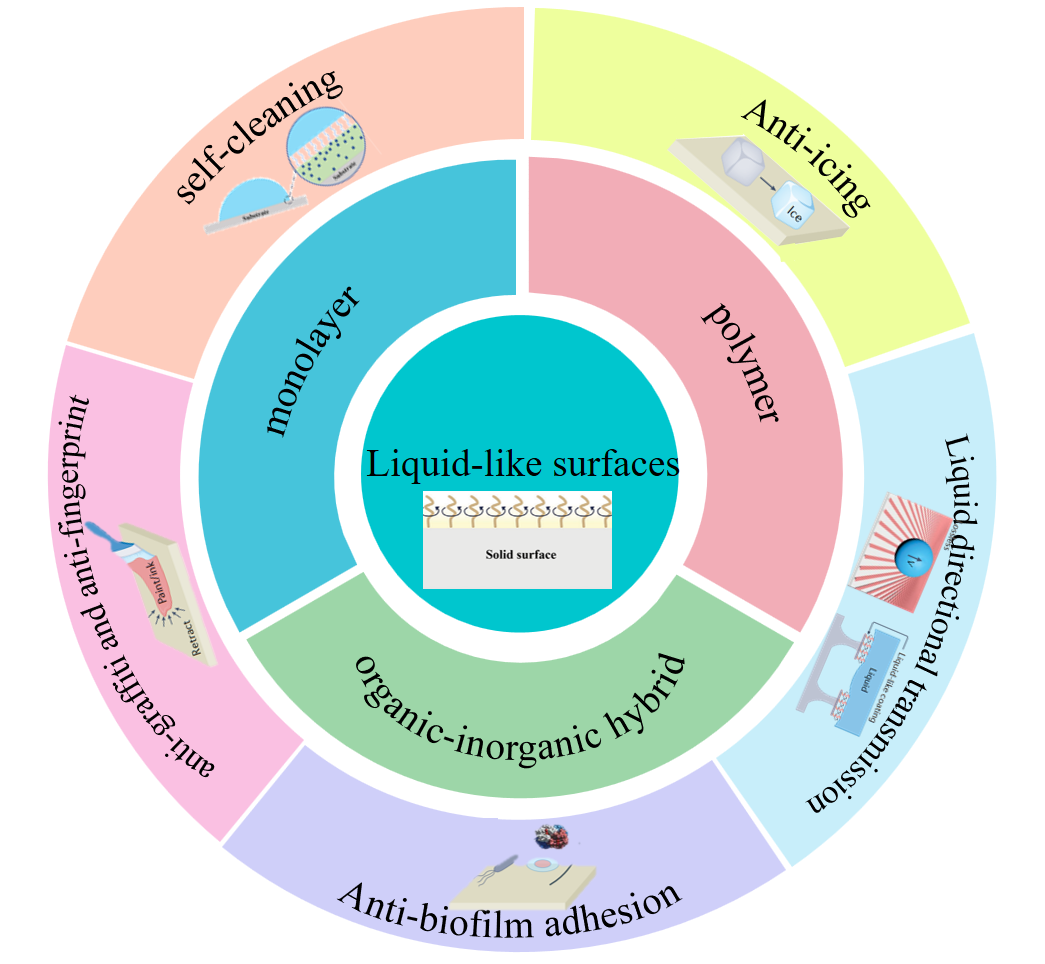
Liquid-like surfaces (LLS), as novel bioinspired interfacial materials, form dynamic molecular brush interfaces through the covalent grafting of flexible polymers or alkyl molecular chains. This approach overcomes the limitations of traditional superhydrophobic surfaces (SHPS) and slippery liquid-infused porous surfaces (SLIPS), which heavily rely on micro/nanostructures or external lubricants. The core advantage of LLS lies in the high mobility of its molecular chains, which significantly reduces contact angle hysteresis (CAH) and sliding angle (SA), enabling droplet self-cleaning at minimal tilt angles or even on horizontal surfaces. This paper first elaborates on the liquid-repellent mechanism of LLS, which involves the use of flexible chains to mask substrate defects and reduce contact line pinning effects, thereby achieving dynamic droplet dewetting. Subsequently, it summarizes the three main types of LLS, including monolayers, polymer layers, and organic-inorganic hybrid layers, and analyzes the relationship between different structures and liquid-repellent performance. Next, the applications of LLS coatings in anti-icing, self-cleaning, graffiti resistance, anti-bioadhesion, directional liquid transport, anti-scaling, and membrane fouling inhibition are reviewed. Finally, the challenges faced by LLS coatings, such as mechanical durability and chemical stability, are discussed, along with future prospects for advancing multifunctional integration.
1 Introduction
2 Mechanism of liquid-like surface
3 The construction method of liquid-like surfaces
3.1 The surface of a liquid-like monolayer
3.2 The surface of liquid-like polymers
3.3 The surface of liquid-like organic-inorganic hybrid
4 Applications on liquid-like surfaces
4.1 Anti-icing
4.2 Self-cleaning
4.3 Anti-fingerprint and anti-graffiti
4.4 Anti-biofilm adhesion
4.5 Liquid directional transmission
4.6 Anti-fouling
4.7 Mitigating membrane fouling
5 Conclusion and outlook
Yan Bao , Chuang Fu , Renhao Li , Wenbo Zhang . Construction Methods and Application Progress of Liquid-Like Surfaces[J]. Progress in Chemistry, 2025 , 37(12) : 1866 -1876 . DOI: 10.7536/PC20250604
图5 类液体表面在防结冰中的应用。(a) HSHC和LSHC涂层的防冰机理图[92];(b) BA、HSHC和LSHC涂层的冰冻结过程效果图[92];(c) BA、HSHC和LSHC涂层冰黏附力的对比图[92];(d) 冰在裸玻璃和光滑涂层表面的黏附强度[93];(e) 冰在光滑涂层上的黏附强度随结冰循环的变化[93]Fig.5 The application of liquid-like surfaces in anti-icing. (a) Anti-icing mechanism diagrams of HSHC and LSHC coatings[92]; (b) effect diagram of the ice freezing process of BA, HSHC and LSHC coatings[92]; (c) comparison chart of ice adhesion force of BA, HSHC and LSHC coatings[92]; (d) the adhesion strength of ice on bare glass and smooth coating surfaces[93]; (e) the variation of the adhesion strength of ice on smooth coatings with the freezing cycle[93] |
图6 类液体表面在自清洁中的应用。(a) 类液体涂层的制备过程示意图[95];(b) 类液体涂层的自清洁效果图[95];(c) MAPOSS-co-PFMA的制备及其侧氟链的部分结晶示意图[96];(d) MAPOSS-co-PFMA涂层低附着力、自愈合、耐高低温和自清洁效果图[96]Fig.6 Applications of liquid-like surfaces in self-cleaning. (a) Schematic diagram of the synthesis of liquid-like coatings[95]; (b) self-cleaning effect diagram of type liquid coating[95]; (c) preparation of MAPOSS-co-PFMA and partial crystallization diagram of its side fluorine chain[96]; (d) effect diagram of low adhesion, self-healing, resistance to high and low temperatures and self-cleaning of MAPOSS-co-PFMA coating[96] |
图7 类液体表面在防涂鸦及耐指纹中的应用。(a) 防涂鸦涂层的形成机理图[97];(b) 在玻璃基材上的防涂鸦效果图[97];(c) 油墨在裸玻璃和经涂层处理的玻璃表面的收缩痕迹和经纸巾擦拭后的效果图[98];(d) 油墨分别在50、100、150、200、250和300次书写和擦除循环后的表面收缩状态[98];(e) 人工指纹液在裸玻璃和经涂层处理的玻璃表面的收缩状态[98]Fig.7 The application of liquid-like surfaces in anti-graffiti and anti-fingerprint properties. (a) Formation mechanism diagram of anti-graffiti coating[97]; (b) anti-graffiti effect drawing on glass substrate[97]; (c) the shrinkage marks of the ink on bare glass and coated glass surfaces and the effect drawing after wiping with paper towels[98]; (d) the surface shrinkage states of the ink after 50, 100, 150, 200, 250 and 300 writing and erasing cycles respectively[98]; (e) the contraction state of artificial fingerprint liquid on bare glass and coated glass surfaces[98] |
图8 类液体表面在抗生物膜黏附中的应用。(a) PQMS、多元醇、HDIT的化学结构式及形成PU-PQMS涂层后对抗生物膜黏附的机理图[101];(b) PU-PDMS和PU-PQMS涂层对大肠杆菌和金黄色葡萄球菌的抗菌效果图[101];(c) PU-PDMS和PU-PQMS涂层对大肠杆菌和金黄色葡萄球菌的抗菌率[101]Fig.8 The application of liquid-like surfaces in anti-biofilm adhesion. (a) The chemical structural formulas of PQMS, polyols and HDIT and the mechanism diagrams of anti-biofilm adhesion after the formation of PU-PQMS coating[101]; (b) antibacterial effect diagrams of PU-PDMS and PU-PQMS coatings on Escherichia coli and Staphylococcus aureus[101]; (c) the antibacterial rates of PU-PDMS and PU-PQMS coatings against Escherichia coli and Staphylococcus aureus[101] |
图9 类液体表面在液体定向传输中的应用。(a) 分别在涂层表面接枝全氟辛基硅烷(刚性结构)和全氟聚醚(柔性结构)示意图[102];(b) 液滴在全氟辛烷硅烷(刚性结构)和全氟聚醚(柔性结构)表面微观运输机理图[102]Fig.9 The application of liquid-like surfaces in liquid directional transmission. (a) Schematic diagram of grafting perfluorooctane silane (rigid structure) and perfluoropolyether (flexible structure) on the coating surface respectively[102]; (b) microscopic transport mechanism diagram of liquid droplets on the surface of perfluorooctane silane (rigid structure) and perfluoropolyether (flexible structure)[102] |
图10 类液体表面在防结垢中的应用。(a) 分别在涂层表面接枝PDMS、全氟烷基硅烷(PFOS)和十八烷基三氯硅烷(OTS)的示意图[104];(b) 涂层表面接枝PDMS、全氟烷基硅烷(PFOS)和十八烷基三氯硅烷(OTS)的防结垢测试效果图[104]Fig.10 Application of liquid-like surfaces in anti-fouling. (a) Schematic diagram of grafting PDMS, perfluoroalkylsilane (PFOS), and octadecyltrichlorosilane (OTS) onto coated surfaces, respectively[104]; (b) anti-fouling test results of coated surfaces grafted with PDMS, perfluoroalkylsilane (PFOS), and octadecyltrichlorosilane (OTS)[104] |
图11 类液体表面在抑制膜污染中的应用。(a) 分别在表面接枝PDMS、交联聚二甲基硅氧烷(CPDMS)、十八烷基三氯硅烷(OTS)和全氟烷基硅烷(PFOS)的示意图[106];(b) 表面接枝PDMS、交联聚二甲基硅氧烷(CPDMS)、十八烷基三氯硅烷(OTS)和全氟烷基硅烷(PFOS)的抑制膜污染测试效果图[106]Fig. 11 Application of liquid-like surfaces in membrane fouling mitigation. (a) Schematic diagram of grafting PDMS, cross-linked polydimethylsiloxane (CPDMS), octadecyltrichlorosilane (OTS), and perfluoroalkylsilane (PFOS) onto surfaces, respectively[106]; (b) membrane fouling inhibition test results of surfaces grafted with PDMS, cross-linked polydimethylsiloxane (CPDMS), octadecyltrichlorosilane (OTS), and perfluoroalkylsilane (PFOS)[106] |
| [1] |
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
(李玥, 卢亚妹, 王鹏飞, 曹莹泽, 戴春爱. 化学进展, 2021, 33(12): 2362)
|
| [13] |
(许宁, 林雨, 雒玉欣, 马家辉, 杨翘宇, 王卓, 蒲永平, 丁旭东. 陕西科技大学学报, 2024, 42(03): 119)
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
(许金凯, 蔡倩倩, 于占江, 廉中旭, 田纪文, 于化东. 化学进展, 2021, 33(6): 958)
|
| [19] |
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
(
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
|
| [36] |
(蓝敏杰, 文庆珍, 朱金华. 材料保护, 2020, 53(03): 129.)
|
| [37] |
|
| [38] |
(韦代东, 李惠枝, 曾娟娟, 赵传国, 李士强. 中国塑料, 2023, 37(02): 15)
|
| [39] |
(
|
| [40] |
(汪雪, 谢晖, 黄莉, 朱小龙, 赵金国, 曾其昕. 南京工业大学学报(自然科学版), 2024, 46(05): 521)
|
| [41] |
(王文静, 曾显华, 刘鹏碧. 广东化工, 2024, 51(20): 30)
|
| [42] |
|
| [43] |
|
| [44] |
|
| [45] |
|
| [46] |
|
| [47] |
|
| [48] |
|
| [49] |
|
| [50] |
|
| [51] |
|
| [52] |
|
| [53] |
|
| [54] |
|
| [55] |
|
| [56] |
|
| [57] |
|
| [58] |
|
| [59] |
|
| [60] |
|
| [61] |
|
| [62] |
|
| [63] |
|
| [64] |
|
| [65] |
|
| [66] |
|
| [67] |
|
| [68] |
|
| [69] |
|
| [70] |
|
| [71] |
|
| [72] |
|
| [73] |
|
| [74] |
|
| [75] |
|
| [76] |
|
| [77] |
|
| [78] |
|
| [79] |
|
| [80] |
|
| [81] |
|
| [82] |
|
| [83] |
|
| [84] |
|
| [85] |
|
| [86] |
|
| [87] |
|
| [88] |
|
| [89] |
|
| [90] |
|
| [91] |
|
| [92] |
|
| [93] |
|
| [94] |
(李承刚, 吴石莲, 常国华, 关润泽, 周炳见, 杨彤, 杨宇. 材料导报, 2024, 38(23): 247)
|
| [95] |
|
| [96] |
|
| [97] |
|
| [98] |
|
| [99] |
(张群利, 崔琳琳, 高雪. 精细化工, 2022, 39(05): 892.)
|
| [100] |
|
| [101] |
|
| [102] |
|
| [103] |
|
| [104] |
|
| [105] |
|
| [106] |
|
| [107] |
|
/
| 〈 |
|
〉 |