

Biochar System for Phosphorus Cycling: Enhanced Recovery from Wastewater and Performance Evaluation of Derived Slow-Release Phosphorus Fertilizers
Received date: 2025-06-20
Revised date: 2025-08-27
Online published: 2025-12-10
Supported by
National Natural Science Foundation of China(52270078)
Xi'an Jiaotong University Fundamental Research Program(xzy022025024)
Shaanxi Provincial Science Fund for Distinguished Young Scholars(2025JC-JCQN-027)
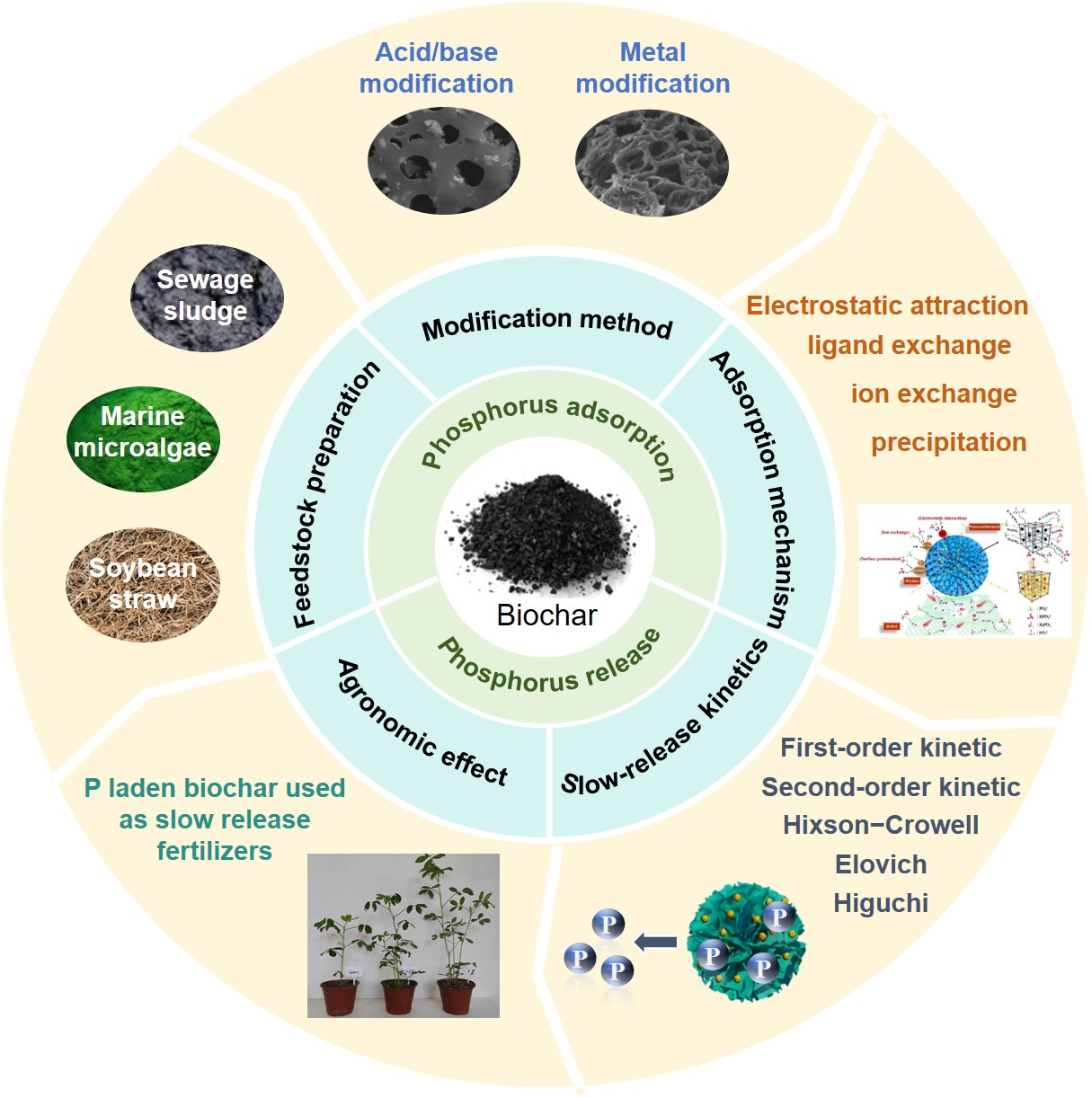
Efficient recovery and recycling of phosphorus are of dual strategic significance to alleviate global phosphorus shortage and eutrophication. As a green, economical and multifunctional porous carbon material, biochar is an ideal carrier for phosphorus recovery and slow-release utilization. This paper discusses the influence of biomass feedstock and pyrolysis process on phosphorus adsorption capacity, and puts forward the principles of feedstock screening and preparation process optimization. Secondly, the metal modification-based enhancement strategy is analyzed in detail, and the mechanism and advantages of metal doping in enhancing phosphorus adsorption performance are clarified. Next, the synergistic effects involving electrostatic attraction, ion exchange, ligand exchange and surface precipitation during biochar phosphorus adsorption are systematically revealed, and functional groups and Lewis acid-base interactions contribute to the selectivity of phosphorus adsorption. The application of slow-release kinetic models to evaluate the phosphorus release mechanism is discussed, and a phosphorus fertilizer efficiency evaluation system is established by integrating slow-release characteristics and agronomic effect assessment. Finally, the future problems and directions are outlined to provide theoretical references for advancing this field.
1 Introduction
2 Strategies for preparation and modification of biochar-based adsorbents
2.1 Feedstock selection and preparation
2.2 Modification strategies for biochar
3 Biochar-phosphorus recovery mechanism
3.1 Synergistic mechanisms
3.2 Selective adsorption mechanisms
4 Performance evaluation of biochar-based phosphate fertilizer
4.1 Evaluation of phosphorus release kinetics and slow release properties
4.2 Assessment of agronomic effects
5 Conclusions and outlook
Yunxian Liu , Xue Zhou , Hao Xu , Wei Yan . Biochar System for Phosphorus Cycling: Enhanced Recovery from Wastewater and Performance Evaluation of Derived Slow-Release Phosphorus Fertilizers[J]. Progress in Chemistry, 2025 , 37(12) : 1902 -1916 . DOI: 10.7536/PC20250614
| Feedstocks | C (%) | N (%) | O (%) | Ash (%) |
|---|---|---|---|---|
| Agriculture | 27~65 | 0.5~4.5 | 17~49 | 0.8~12.6 |
| Aquatic | 38~54 | 1~12 | 26~53 | 13~43 |
| Food | 44~53 | 0.5~1.3 | 34~46 | 0.3~8.1 |
| Forest | 49~58 | 2~3 | 29~38 | 5~11 |
| Industrial | 78~80 | 11~15 | 20~48 | 0.5~7.8 |
表2 不同改性方法在磷吸附方面的适用条件、优势和局限Table 2 Applicable conditions, advantages, and limitations of different modification methods in phosphorus adsorption |
| Modification Method | Applicable Conditions | Advantages | Limitations |
|---|---|---|---|
| Acid Modification | Effective in acidic to neutral conditions. | 1. Introduces acidic functional groups (e.g., —COOH, —OH). 2. Improves pore structure.3. Simple and low-cost. | 1. Efficiency declines sharply in alkaline conditions. 2. Limited adsorption capacity and selectivity. 3. Strong acids are hazardous and may degrade pore structure. |
| Alkali Modification | Effective in neutral to alkaline conditions. | 1. Significantly increases surface area and porosity. 2. Serves as an effective pre-treatment for further modification. | 1. Provides limited direct enhancement of phosphate adsorption. 2. Often requires additional modification to achieve significant results. 3. Strong alkalis are hazardous to handle. |
| Metal salt Modification | Broad pH range (depends on metal type) | 1. Highly effective through strong chemical reactions. 2. High adsorption capacity and selectivity. 3. Recovered phosphate products can be utilized as fertilizer. | 1. Relatively high cost, especially for rare-earth metals (e.g., La). 2. Potential metal leaching leading to secondary pollution. 3. Excessive metal loading may clog pores. |
| Multi-metal Modification | Broadest pH range and adaptability. | 1. Synergistic effects enhance overall performance. 2. Incorporation of low-cost metals helps reduce expenses. 3. Generally superior to single-metal modified biochar. | 1. Preparation process is more complex. 2. Interaction mechanisms between metals are not fully understood. |
图4 (A) Mg@ABB的DFT计算[61];不同材料的SEM图对比:(B) 原始玉米芯生物炭[62];(C) CaCl2改性玉米芯生物炭[62];(D) 原始荞麦壳生物炭[64];(E) CaCl2改性荞麦壳生物炭[64]Fig.4 (A) DFT calculations of Mg@ABB[61]; comparison of SEM images of different materials: (B) pristine corn cob biochar[62]; (C) CaCl2-modified corn cob biochar[62]; (D) pristine buckwheat hull biochar[64]; (E) CaCl2-modified buckwheat hull biochar[64] |
图5 基于Langmuir模型的金属基吸附剂最大吸附量与伪二级动力学系数的比较:Mg-SCG-500[74],Mg-SCG-600[74],SA-KBC-Fe/La[75],Fe-MnBC[76],Fe-CNT-2[77],MBA/AlCl3[78],La-Spinel/Biochar[79],NCo-CA[80],Mg@CaO composite biochar[81],MPG-BC 1:1[82],Fe-CaBC[83],La3.5-CA-450[84]Fig.5 Comparison of the maximum adsorption capacity from the Langmuir model and pseudo-second order kinetic coefficients of reported metal-based adsorbents: Mg-SCG-500[74], Mg-SCG-600[74], SA-KBC-Fe/La[75], Fe-MnBC[76], Fe-CNT-2[77], MBA/AlCl3[78], La-Spinel/Biochar[79], NCo-CA[80], Mg@CaO composite biochar[81], MPG-BC 1:1[82], Fe-CaBC[83], La3.5-CA-450[84] |
表3 原始生物炭和改性生物炭对磷酸盐的吸附研究Table 3 Adsorption studies of phosphate on virgin and modified biochar |
| Feedstocks | Modification | Initial phosphorus concentration (mg/L) | Maximum adsorption capacity (mg PO43-/g) | Specific surface area (m2/g) | Functional group | Adsorption isotherm | Adsorption mechanism | Ref |
|---|---|---|---|---|---|---|---|---|
| Crawfish char | - | 2~240 | 70.6 | - | CO32-,―OH | Langmuir/Freundlich | Ion exchange, surface precipitation | 47 |
| Pineapple | - | 100~400 | 3.9 | 32.22 | ―OH,―CH, C―O | Langmuir | Ion exchange, surface precipitation | 48 |
| Eupatorium adenophorum | - | 20~300 | 13.6 | 65.99 | CO32-,―OH, ―CH,C―O | Langmuir | - | 49 |
| Anaerobic digestion residues | MgO,Fe2O3 | 200~1000 | 149.25 | 17.53 | Fe―O,Mg―O, C | Langmuir | Physical adsorption, precipitation, surface complexation, electrostatic attraction | 50 |
| Mushroom waste | MgCl2,Fe2O3 | 30 | 247 | 396.01 | Fe―O,MgOH+,FeOH+,M―OH,―OH,―COOH | Langmuir | Ion exchange, precipitation, electrostatic attraction | 51 |
| Bagasse | Marble waste | 100 | 261.37 | 92.81 | Mg―O, Mg―OH,―OH | Langmuir | Electrostatic attraction, co‑precipitation | 52 |
| Rape straw | Eggshells | 10~200 | 109.7 | 181.32 | C C | Langmuir | Hydrogen bonding, electrostatic attraction, precipitation | 53 |
| Potato peels | Eggshells | 5~250 | 174.8 | - | CO32-,―OH | Langmuir | Chemical adsorption, precipitation | 54 |
| Wheat straw | Nano‑CaO2 | 100~400 | 213.22 | 1.12 | ―OH,C | Langmuir | Precipitation | 55 |
| Corn stalks | NaLa(CO3)2,Fe3O4 | 50~400 | 330.86 | 20.61 | La(CO3)2-, La(HCO3)+ | Langmuir | Electrostatic attraction, ligand exchange, inner sphere complexation | 56 |
| Canna | La(OH)3,Fe2O3 | 20~500 | 101.16 | 84.89 | O―P―O, ―OH,O2- | Langmuir | Electrostatic attraction, ion exchange, ligand exchange, inner‑sphere complexation | 57 |
表4 磷缓释动力学模型及相关机理Table 4 Evaluation models for kinetics and mechanism of phosphorus slow release |
| Model | Mathematical expression | Parameter description | Scope of application |
|---|---|---|---|
| Elovich | | Qt is the release concentration at time t;α is the initial release rate;β is the rate change factor | Phosphorus adsorption and desorption as influenced by the surface coverage of fertilizer particles; applicable to phosphorus adsorption and desorption processes |
| First-order | ln | Mt and M∞ are the percentage of phosphorus released at different times, respectively. k1 is the first-order release rate constant | The relationship between phosphorus concentration and time is assumed to be linear, and the rate of phosphorus release depends on the initial fertilizer concentration |
| Second-order kinetics | | Qt and Qmax are the phosphorus release concentrations at time t and equilibrium, respectively; k2 is the second-order release rate constant | Chemically dominated phosphorus release process where the rate of phosphorus release depends on the initial fertilizer concentration |
| Higuchi kinetics | | Mt and M∞ are the percentage of phosphorus released at different times, respectively. k3 is the Higuchi release rate constant | The initial concentration of phosphorus released into solution is much higher than the solubility, and the diffusion of phosphorus occurs in only one dimension; the phosphorus particles are much smaller than the thickness of the system, and edge effects and matrix solubilization are negligible |
| Hixson-Crowell | | Mt and M∞ are the percentage of phosphorus released at different times, respectively. k4 is the Hixson-Crowell release rate constant | Release process dominated by dissolution of phosphorus particles; dissolution is assumed to occur at the surface of the fertilizer particles, with sufficient agitation and constant particle shape |
| [1] |
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
(赵健慧, 李欣欣, 李欢, 常晶, 李宁, 王少坡. 净水技术, 2024, 43(6): 44, 70.)
|
| [14] |
(王书燕, 张新波, 彭安萍, 刘阳,
|
| [15] |
(梁文洁, 郭盼梁, 石晟昊, 梁海. 山东化工, 2022, 51(6): 137)
|
| [16] |
|
| [17] |
|
| [18] |
|
| [19] |
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
|
| [36] |
|
| [37] |
|
| [38] |
|
| [39] |
|
| [40] |
(张雨禾, 庄舜尧. 环境污染与防治, 2020, 42(10): 1216)
|
| [41] |
|
| [42] |
|
| [43] |
(朱艳, 肖清波, 奚永兰, 高娣, 王宇欣, 杜静, 叶小梅. 生态环境学报, 2020, 29(9): 1897)
|
| [44] |
(唐勇, 吴桐, 勾曦, 王波, 罗婷, 谢燕华. 水处理技术, 2021, 47(11): 91)
|
| [45] |
|
| [46] |
|
| [47] |
|
| [48] |
|
| [49] |
|
| [50] |
|
| [51] |
|
| [52] |
|
| [53] |
|
| [54] |
|
| [55] |
|
| [56] |
|
| [57] |
|
| [58] |
|
| [59] |
|
| [60] |
|
| [61] |
|
| [62] |
|
| [63] |
(常思露, 高茜, 魏佳宇, 刘凤旭, 阎波, 王文华. 环境工程学报, 2024, 18(2): 481)
|
| [64] |
|
| [65] |
|
| [66] |
(纳云, 马巍, 李娜, 刘陆, 陈荣志. 水处理技术, 2024, 50(11): 31.)
|
| [67] |
|
| [68] |
|
| [69] |
|
| [70] |
|
| [71] |
|
| [72] |
|
| [73] |
|
| [74] |
|
| [75] |
|
| [76] |
|
| [77] |
|
| [78] |
|
| [79] |
|
| [80] |
|
| [81] |
|
| [82] |
|
| [83] |
|
| [84] |
|
| [85] |
|
| [86] |
|
| [87] |
|
| [88] |
|
| [89] |
|
| [90] |
|
| [91] |
|
| [92] |
|
| [93] |
|
| [94] |
|
| [95] |
|
| [96] |
|
| [97] |
|
| [98] |
|
| [99] |
|
| [100] |
|
| [101] |
|
| [102] |
|
| [103] |
|
| [104] |
|
| [105] |
|
| [106] |
|
| [107] |
|
| [108] |
|
| [109] |
|
| [110] |
|
| [111] |
|
| [112] |
|
| [113] |
|
| [114] |
|
| [115] |
|
| [116] |
|
| [117] |
|
| [118] |
|
| [119] |
|
| [120] |
|
| [121] |
|
| [122] |
|
| [123] |
|
| [124] |
|
| [125] |
|
| [126] |
|
/
| 〈 |
|
〉 |