

Synthesis of Graphynes and Their Applications in Third-Order Nonlinear Optics
Received date: 2025-05-26
Revised date: 2025-07-02
Online published: 2025-12-10
Supported by
National Natural Science Foundation of China(22275202)
Natural Science Foundation of Shanxi Province(20210302123144)
National Natural Science Foundation of China(21875267)
Shanxi Scholarship Council of China(2021-057)
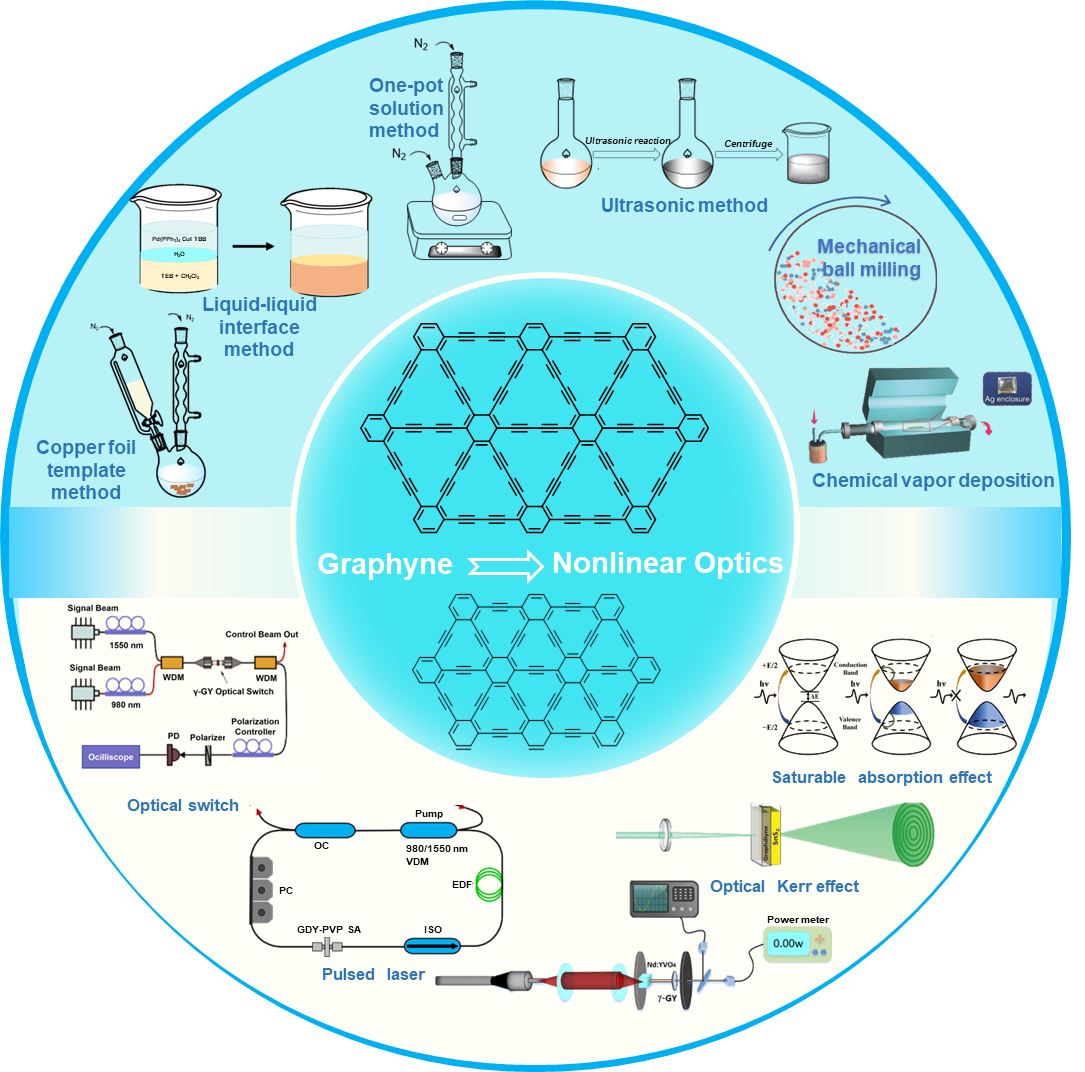
Graphynes are a kind of low-dimensional carbon material composed of sp- and sp²-hybridized carbon atoms with unique electronic conjugation topologies and tunable chemical properties. Recently, significant progress has been made in the synthesis methods of graphynes. Various derivative structures as well as different morphologies from nanosheets to macroscopic films have been achieved through dry or wet chemical methods, which provide important theoretical and experimental supports for designing new carbon materials. Due to the high specific surface areas, abundant chemically active sites, and adjustable bandgap structures, graphyne derivatives exhibit high nonlinear optical coefficients and ultra-fast carrier migration rates, revealing great application potential in nonlinear optics. In this paper, the structural classification, synthesis strategies, and third-order nonlinear optical properties of graphynes are systematically reviewed, aiming to provide references for practical applications of graphynes in optical and optoelectronic fields.
1 Introduction
2 Structure of Graphyne
2.1 Structure of intrinsic Graphyne
2.2 Structure of Graphyne derivatives
3 Preparation of graphyne carbon materials
3.1 Preparation of graphdiynes
3.2 Preparation of graphynes
4 Applications of graphynes in third‑order nonlinear optics
4.1 Optical Kerr effect
4.2 Saturable absorption
4.3 Reverse saturable absorption
5 Conclusion and outlook
Key words: graphyne; graphyne derivatives; nonlinear optics; optoelectionic devices
Juemin Zhao , Bin Liang , Yaxing Tang , Jie Li , Zheng Xie . Synthesis of Graphynes and Their Applications in Third-Order Nonlinear Optics[J]. Progress in Chemistry, 2025 , 37(12) : 1917 -1930 . DOI: 10.7536/PC20250520
图3 (a) 化学气相沉积法制备GDY[51];(b) 化学气相沉积法制备F-GDY/H-GDY[52];(c) F-GDY的AFM和HRTEM图像[53];(d) ZnO纳米阵列上沉积GDY薄膜示意图和(e) TEM和HRTEM图像[54]Fig.3 (a) Schematic diagram of GDY synthesized by CVD method[51]; (b) schematic diagram of F-GDY/H-GDY synthesized by CVD method[52]; (c) AFM and HRTEM images of F-GDY[53]; (d) schematic diagram of GDY thin films deposited on ZnO nano-arrays; (e) TEM and HRTEM images of GDY thin films[54] |
图4 (a) 铜箔生长法合成GDY反应流程图及SEM和TEM图像[11];(b) 四苯乙烯单元的荧光石墨炔衍生物薄膜的合成示意图[39];(c) OMe-GDY合成路线图[56];(d) TzlGDY合成路线、HRTEM和SAED图像[57]Fig.4 (a) Synthetic route, SEM and TEM images of GDY based on copper foil method[11]; (b) schematic diagram of the synthesis of fluorescent TPE-GDY[39]; (c) synthetic route to OMe-GDY[56]; (d) synthetic route, HRTEM and SAED images of TzlGDY[57] |
图5 (a) GDY液-液/气-液界面法示意图及电镜表征结果[58];(b) 两种拓扑结构TPE-GDY的液/液界面法示意图[59];(c) GDY纳米线合成示意图及电镜表征结果[60];(d) 固/液界面温度梯度驱动合成GDY[61]Fig.5 (a) Schematic diagram and electron microscope characterization of GDY by liquid-liquid/gas-liquid interfacial method[58]; (b) schematic diagram of liquid/liquid interface method for two topologies of TPE-GDY[59]; (c) schematic diagram and electron microscopy characterization of GDY nanowires[60]; (d) temperature gradient-driven synthesis of GDY at solid/liquid interfaces[61] |
图7 (a) 机械球磨法合成H-GY[63];(b) 机械球磨法合成GY[64];(c) GY的拉曼光谱和粉末照片[64];(d) CVD法合成GY反应示意图及(e)HRTEM图谱(插图为SAED图谱)[66]Fig.7 (a) Synthesis of H-GY by mechanical ball milling[63]; (b) synthesis of GY by mechanical ball milling[64]; (c) Raman spectrum and powder photographs of GY[64]; (d) schematic illustration of the synthesis of GY by CVD method and (e) HRTEM images (inset are SAED images)[66] |
图11 (a) GDY/SnS2光子二极管示意图[83];(b) GDY在不同溶剂中归一化透射率的Z扫描曲线和理论拟合[17]Fig.11 (a) Schematic diagram of non-reciprocal light propagation phenomena in GDY/SnS2 photodiode[83]; (b) experimental results and theoretical fits of short aperture Z-scan curves of normalized transmittance of GDY dispersions in different solvents[17] |
图12 (a) GDY非线性传输和拟合曲线;GDY锁模光纤激光器的(b)输出脉冲串和(c)光谱学[88];GDY SA光纤器件在(d)1.5 µm,(e) 2 µm处的非线性能量依赖传输曲线[89];(f) GDY-PMMA混合锁模光纤激光器的自相关轨迹[90];(g, h) GDY SA光纤激光器的输出频谱及可调谐脉冲激光器的输出光谱[91]Fig.12 (a) Nonlinear transmission and fitting curves of GDY; (b) output pulse train and (c) spectroscopy of a GDY based mode-locked fiber laser[88]; nonlinear energy-dependent transmission curves of a GDY SA fiber device (d) at 1.5 µm; (e) at 2 µm[89]; (f) autocorrelation trajectories of a GDY-PMMA hybrid mode-locked fiber laser[90]; (g, h) output spectrum, single-pulse curves and tunable-pulse laser output spectra of a GDY output spectra of SA fiber laser and tunable pulse laser[91] |
图13 (a) GY SA的CW和Q开关激光脉冲的光谱;(b)GY SA的自相关轨迹[92];(c) HgL1为SA的Q开关单脉冲能量和峰值功率;(d) HgL2为SA的Q开关单脉冲能量和峰值功率[37]Fig.13 (a) Spectra of CW and Q-switched laser pulses of GY SA; (b) autocorrelation trajectory of GY SA[92]; (c) HgL1 is the Q-switched single pulse energy and peak power of the saturable absorber; (d) HgL2 is the Q-switched single pulse energy and peak power of the saturable absorber[37] |
图14 (a) GY光学开关设备示意图;(b) GY光学开关的长期输出波形;(c) 信号输入与GY光学开关结果的比较图[92];(d) GDYO的化学结构式;(e) 基于SSPM的GDYO NS的非线性行为;(f) 基于SXPM的GDYO NS的非线性行为[94]Fig.14 (a) Schematic of the GY optical switch; (b) long-term output waveform of the GY optical switch; (c) plot comparing the signal input with the GY optical switching results[92]; (d) synthesis of GDYO; (e) Nonlinear behavior of the GDYO NS based on the SSPM; and (f) nonlinear behavior of the GDYO NS based on the SXPM[94] |
| [1] |
|
| [2] |
|
| [3] |
|
| [4] |
Tahir,
|
| [5] |
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
|
| [19] |
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
|
| [36] |
|
| [37] |
|
| [38] |
|
| [39] |
|
| [40] |
|
| [41] |
|
| [42] |
|
| [43] |
|
| [44] |
|
| [45] |
|
| [46] |
|
| [47] |
|
| [48] |
|
| [49] |
|
| [50] |
|
| [51] |
|
| [52] |
|
| [53] |
|
| [54] |
|
| [55] |
|
| [56] |
|
| [57] |
|
| [58] |
|
| [59] |
|
| [60] |
|
| [61] |
|
| [62] |
|
| [63] |
(李乔丹, 李永, 唐佳宁, 崔晓莉. 物理化学学报, 2018, 34(9): 1080)
|
| [64] |
|
| [65] |
|
| [66] |
|
| [67] |
|
| [68] |
|
| [69] |
|
| [70] |
|
| [71] |
|
| [72] |
|
| [73] |
|
| [74] |
|
| [75] |
|
| [76] |
|
| [77] |
|
| [78] |
|
| [79] |
|
| [80] |
|
| [81] |
|
| [82] |
|
| [83] |
|
| [84] |
|
| [85] |
|
| [86] |
|
| [87] |
|
| [88] |
|
| [89] |
|
| [90] |
|
| [91] |
|
| [92] |
|
| [93] |
|
| [94] |
|
| [95] |
|
| [96] |
|
| [97] |
|
/
| 〈 |
|
〉 |